The protective effects of Xuebijing injection on intestinal injuries of mice exposed to irradiation
- PMID: 36376997
- PMCID: PMC9773304
- DOI: 10.1002/ame2.12285
The protective effects of Xuebijing injection on intestinal injuries of mice exposed to irradiation
Abstract
Background: Gastrointestinal (GI) injury is one of the most common side effects of radiotherapy. However, there is no ideal therapy method except for symptomatic treatment in the clinic. Xuebijing (XBJ) is a traditional Chinese medicine, used to treat sepsis by injection. In this study, the protective effects of XBJ on radiation-induced intestinal injury (RIII) and its mechanism were explored.
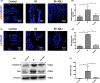
Methods: The effect of XBJ on survival of irradiated C57BL/6 mice was monitored. Histological changes including the number of crypts and the length of villi were evaluated by H&E. The expression of Lgr5+ intestinal stem cells (ISCs), Ki67+ cells, villin and lysozymes were examined by immunohistochemistry. The expression of cytokines in the intestinal crypt was detected by RT-PCR. DNA damage and apoptosis rates in the small intestine were also evaluated by immunofluorescence.
Results: In the present study, XBJ improved the survival rate of the mice after 8.0 and 9.0 Gy total body irradiation (TBI). XBJ attenuated structural damage of the small intestine, maintained regenerative ability and promoted proliferation and differentiation of crypt cells, decreased apoptosis rate and reduced DNA damage in the intestine. Elevation of IL-6 and TNF-α was limited, but IL-1, TNF-𝛽 and IL-10 levels were increased in XBJ-treated group after irradiation. The expression of Bax and p53 were decreased after XBJ treatment.
Conclusions: Taken together, XBJ provides a protective effect on RIII by inhibiting inflammation and blocking p53-related apoptosis pathway.
Keywords: Apoptosis; Inflammation; Intestinal injury; Total body irradiation; Xuebijing injection.
© 2022 The Authors. Animal Models and Experimental Medicine published by John Wiley & Sons Australia, Ltd on behalf of The Chinese Association for Laboratory Animal Sciences.
Conflict of interest statement
There are no conflicts of interest to declare. Aimin Meng is an Editorial Board member of AMEM and a co‐author of this article. To minimize bias, she was excluded from all editorial decision‐making related to the acceptance of this article for publication.
Figures
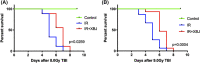




Similar articles
-
Mitigating the effects of Xuebijing injection on hematopoietic cell injury induced by total body irradiation with γ rays by decreasing reactive oxygen species levels.Int J Mol Sci. 2014 Jun 12;15(6):10541-53. doi: 10.3390/ijms150610541. Int J Mol Sci. 2014. PMID: 24927144 Free PMC article.
-
Xuebijing injection reduces organ injuries and improves survival by attenuating inflammatory responses and endothelial injury in heatstroke mice.BMC Complement Altern Med. 2015 Feb 5;15:4. doi: 10.1186/s12906-015-0519-5. BMC Complement Altern Med. 2015. PMID: 25653103 Free PMC article.
-
Anti-sepsis protection of Xuebijing injection is mediated by differential regulation of pro- and anti-inflammatory Th17 and T regulatory cells in a murine model of polymicrobial sepsis.J Ethnopharmacol. 2018 Jan 30;211:358-365. doi: 10.1016/j.jep.2017.10.001. Epub 2017 Oct 4. J Ethnopharmacol. 2018. PMID: 28987599
-
The current evidence for the treatment of sepsis with Xuebijing injection: Bioactive constituents, findings of clinical studies and potential mechanisms.J Ethnopharmacol. 2021 Jan 30;265:113301. doi: 10.1016/j.jep.2020.113301. Epub 2020 Aug 27. J Ethnopharmacol. 2021. PMID: 32860891 Review.
-
The effects of Xuebijing injection combined with ulinastatin as adjunctive therapy on sepsis: An overview of systematic review and meta-analysis.Medicine (Baltimore). 2022 Oct 21;101(42):e31196. doi: 10.1097/MD.0000000000031196. Medicine (Baltimore). 2022. PMID: 36281160 Free PMC article.
Cited by
-
Establishment and evaluation of an improved rat model of open abdomen.Animal Model Exp Med. 2024 Aug;7(4):562-569. doi: 10.1002/ame2.12376. Epub 2023 Dec 29. Animal Model Exp Med. 2024. PMID: 38158631 Free PMC article.
-
The Natural Product Secoemestrin C Inhibits Colorectal Cancer Stem Cells via p38-S100A8 Feed-Forward Regulatory Loop.Cells. 2024 Apr 3;13(7):620. doi: 10.3390/cells13070620. Cells. 2024. PMID: 38607060 Free PMC article.
-
Development of radiation countermeasure agents for acute radiation syndromes.Animal Model Exp Med. 2023 Aug;6(4):329-336. doi: 10.1002/ame2.12339. Epub 2023 Aug 29. Animal Model Exp Med. 2023. PMID: 37642199 Free PMC article. Review.
-
Effects of Bacillus halophilus on growth, intestinal flora and metabolism of Larimichthys crocea.Biochem Biophys Rep. 2023 Sep 12;35:101546. doi: 10.1016/j.bbrep.2023.101546. eCollection 2023 Sep. Biochem Biophys Rep. 2023. PMID: 37731665 Free PMC article.
References
-
- Xu Y, Tu W, Sun D, et al. Nrf2 alleviates radiation‐induced rectal injury by inhibiting of necroptosis. Biochem Biophys Res Commun. 2021;554:49‐55. - PubMed
-
- Monti P, Wysocki J, Van der Meeren A, Griffiths NM. The contribution of radiation‐induced injury to the gastrointestinal tract in the development of multi‐organ dysfunction syndrome or failure. BJR Suppl BIR. 2005;27:89‐94.
-
- Paquette IM, Vogel JD, Abbas MA, Feingold DL, Steele SR, Colon CPGCoTASo, Rectal S . The American Society of Colon and Rectal Surgeons Clinical Practice guidelines for the treatment of chronic radiation proctitis. Dis Colon Rectum. 2018;61(10):1135‐1140. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

