Bimatoprost Implant Biodegradation in the Phase 3, Randomized, 20-Month ARTEMIS Studies
- PMID: 36378864
- PMCID: PMC9885540
- DOI: 10.1089/jop.2022.0137
Bimatoprost Implant Biodegradation in the Phase 3, Randomized, 20-Month ARTEMIS Studies
Abstract
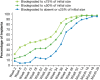
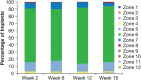
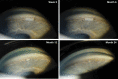
Purpose: To evaluate the time course of biodegradation of an intracameral, biodegradable, sustained-release bimatoprost implant that lowers intraocular pressure without the need for daily eye drops. Methods: In 2 identically designed, randomized, phase 3 clinical trials, adults with open-angle glaucoma or ocular hypertension and open iridocorneal angles inferiorly in the study eye were administered 10- or 15-μg bimatoprost implant (day 1 and weeks 16 and 32) or twice-daily topical timolol 0.5%. Implants were assessed on gonioscopy throughout the studies. Investigators reported whether implants were visible, estimated the size of visible implants relative to their initial size at implantation, and reported the implant location. Data for 10-μg implant placed on day 1 were pooled from both studies for analysis. Results: A total of 372 patients received the 10-μg bimatoprost implant. The degree of implant biodegradation at each follow-up time point was variable among patients. The implant frequently swelled during the initial phase of biodegradation from 6 to 28 weeks. Accelerated biodegradation occurred between 31 and 52 weeks, resulting in 82% of implants absent or ≤25% of initial size by 52 weeks. By month 20, 95% of implants had biodegraded to absent or ≤25% of initial size. The implant was predominantly located inferiorly in the iridocorneal angle. Conclusions: Bimatoprost implant biodegradation in phase 3 studies showed some degree of variability among patients. Clinically significant implant biodegradation was observed in the majority of patients by 12 months. Clinical studies are in progress to further understand implant biodegradation and the ideal timing for implant re-administration. ClinicalTrials.gov NCT02247804; ClinicalTrials.gov NCT02250651.
Keywords: biodegradable implant; drug delivery system; intraocular injection; prostaglandin analog; randomized clinical trial.
Conflict of interest statement
Robert N. Weinreb: consultant for AbbVie, Aerie Pharmaceuticals, Allergan, Equinox, Eyenovia, Iantrek, Implandata, IOPtic, Nicox, and Topcon; research support from Bausch + Lomb, Carl Zeiss Meditec, Centervue, Heidelberg Engineering, National Eye Institute, National Institute of Minority Diseases and Health Disparities, Optovue, Topcon, and Zilia; and founder of Toromedes.
Jason Bacharach: consultant and on the speaker's bureau for, and receives research support from, Allergan (an AbbVie company).
Jacob W. Brubaker: consultant for and has received research support from Allergan (an AbbVie company), Aerie Pharmaceuticals, Equinox, Glaukos, Alcon, and Santen; consultant for Iridex and New World Medical; and receives research support from Nicox and Twenty/Twenty Therapeutics.
Felipe A. Medeiros: consultant for Aerie Pharmaceuticals, Allergan (an AbbVie company), Annexon, Biogen, Carl Zeiss Meditec, Galimedix, Novartis, Reichert, and Stealth BioTherapeutics; research support from Carl Zeiss Meditec, Google, Heidelberg Engineering, and Reichert; and founder of NGoggle, Inc.
Marina Bejanian and Michael R. Robinson: employee of AbbVie, Inc.
Paula Bernstein: contractor for AbbVie, Inc.
Figures



References
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
