Roles of effective stabilizers in improving oral bioavailability of naringenin nanocrystals: Maintenance of supersaturation generated upon dissolution by inhibition of drug dimerization
- PMID: 36382308
- PMCID: PMC9640469
- DOI: 10.1016/j.ajps.2022.09.001
Roles of effective stabilizers in improving oral bioavailability of naringenin nanocrystals: Maintenance of supersaturation generated upon dissolution by inhibition of drug dimerization
Abstract
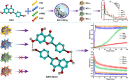
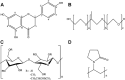
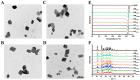
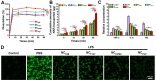
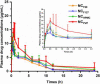
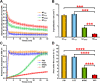
Nanocrystals (NCs), a colloidal dispersion system formulated with stabilizers, have attracted widespread interest due to their ability to effectively improve the oral bioavailability of poorly water-soluble drugs. The stabilizer plays a key role because it can affect the physical stability and even the oral bioavailability of NCs. However, how stabilizers affect the bioavailability of NCs remains unknown. In this study, F68, F127, HPMC, and PVP were each used as a stabilizer to formulate naringenin NCs. The NCs formulated with PVP exhibited excellent release behaviors, cellular uptake, permeability, oral bioavailability, and anti-inflammatory effects. The underlying mechanism is that PVP effectively inhibits the formation of naringenin dimer, which in turn improves the physical stability of the supersaturated solution generated when NC is dissolved. This finding provides insights into the effects of stabilizers on the in vivo performances of NCs and supplies valuable knowledge for the development of poorly water-soluble drugs.
Keywords: Bioavailability; Dimer; Nanocrystals; Stabilizers; Supersaturation maintenance.
© 2022 Shenyang Pharmaceutical University. Published by Elsevier B.V.
Conflict of interest statement
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of this article.
Figures

Similar articles
-
Spironolactone nanocrystals for oral administration: Different pharmacokinetic performances induced by stabilizers.Colloids Surf B Biointerfaces. 2016 Nov 1;147:73-80. doi: 10.1016/j.colsurfb.2016.07.051. Epub 2016 Jul 28. Colloids Surf B Biointerfaces. 2016. PMID: 27490456
-
Stabilizer-induced different in vivo behaviors for intramuscularly long-acting celecoxib nanocrystals.Int J Pharm. 2022 Nov 25;628:122298. doi: 10.1016/j.ijpharm.2022.122298. Epub 2022 Oct 17. Int J Pharm. 2022. PMID: 36257466
-
Supersaturation of zafirlukast in fasted and fed state intestinal media with and without precipitation inhibitors.Eur J Pharm Sci. 2016 Aug 25;91:31-9. doi: 10.1016/j.ejps.2016.05.026. Epub 2016 May 31. Eur J Pharm Sci. 2016. PMID: 27260089
-
Nanocrystals of Poorly Soluble Drugs: Drug Bioavailability and Physicochemical Stability.Pharmaceutics. 2018 Aug 21;10(3):134. doi: 10.3390/pharmaceutics10030134. Pharmaceutics. 2018. PMID: 30134537 Free PMC article. Review.
-
Stabilizing Agents for Drug Nanocrystals: Effect on Bioavailability.Pharmaceutics. 2016 May 20;8(2):16. doi: 10.3390/pharmaceutics8020016. Pharmaceutics. 2016. PMID: 27213435 Free PMC article. Review.
Cited by
-
Solubilization techniques used for poorly water-soluble drugs.Acta Pharm Sin B. 2024 Nov;14(11):4683-4716. doi: 10.1016/j.apsb.2024.08.027. Epub 2024 Sep 2. Acta Pharm Sin B. 2024. PMID: 39664427 Free PMC article. Review.
-
Supersaturation and Precipitation Applicated in Drug Delivery Systems: Development Strategies and Evaluation Approaches.Molecules. 2023 Feb 27;28(5):2212. doi: 10.3390/molecules28052212. Molecules. 2023. PMID: 36903470 Free PMC article. Review.
-
Mechanistic Insight into the Enhanced Anti-Pulmonary Hypertension Efficacy of Wogonin Co-Amorphous.Pharmaceutics. 2025 May 30;17(6):724. doi: 10.3390/pharmaceutics17060724. Pharmaceutics. 2025. PMID: 40574037 Free PMC article.
-
Cationic Curcumin Nanocrystals Liposomes for Improved Oral Bioavailability: Formulation Development, Optimization, In Vitro and In Vivo Evaluation.Pharmaceutics. 2024 Aug 31;16(9):1155. doi: 10.3390/pharmaceutics16091155. Pharmaceutics. 2024. PMID: 39339192 Free PMC article.
-
Spanlastic Nano-Vesicles: A Novel Approach to Improve the Dissolution, Bioavailability, and Pharmacokinetic Behavior of Famotidine.Pharmaceuticals (Basel). 2024 Nov 29;17(12):1614. doi: 10.3390/ph17121614. Pharmaceuticals (Basel). 2024. PMID: 39770456 Free PMC article.
References
-
- Elz A.S., Trevaskis N.L., Porter C.J.H., Bowen J.M., Prestidge C.A. Smart design approaches for orally administered lipophilic prodrugs to promote lymphatic transport. J Control Release. 2022;341:676–701. - PubMed
-
- Homayouni A., Amini M., Sohrabi M., Varshosaz J., Nokhodchi A. Curcumin nanoparticles containing poloxamer or soluplus tailored by high pressure homogenization using antisolvent crystallization. Int J Pharm. 2019;562:124–134. - PubMed
-
- Naseema A., Kovooru L., Behera A.K., Kumar K.P.P., Srivastava P. A critical review of synthesis procedures, applications and future potential of nanoemulsions. Adv Colloid Interface Sci. 2021;287 - PubMed
LinkOut - more resources
Full Text Sources

