Histopathological Features of Cancer-Associated Venous Thromboembolism: Presence of Intrathrombus Cancer Cells and Prothrombotic Factors
- PMID: 36384269
- PMCID: PMC9762717
- DOI: 10.1161/ATVBAHA.122.318463
Histopathological Features of Cancer-Associated Venous Thromboembolism: Presence of Intrathrombus Cancer Cells and Prothrombotic Factors
Abstract
Background: Cancer-associated venous thromboembolism (VTE) is a critical complication in patients with cancer. However, the pathological findings of VTE are limited. Here, we investigated the histopathological features of cancer-associated VTE in human autopsy cases.
Methods: We clinically examined the autopsy cases of VTE with (n=114) and without cancer (n=66) and immunohistochemically analyzed the expression of prothrombotic factors in intrathrombus cancer cells, the thrombus contents of erythrocytes, fibrin, platelets, citrullinated histone H3, and degree of organization.
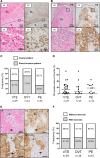
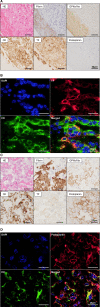
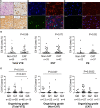
Results: Vascular wall invasion or small cell clusters of cancer cells was observed in thrombi in 27.5% of deep vein thrombosis and 25.9% of pulmonary embolism cases. The majority of the cancer cells in deep vein thrombi appeared to be invading the vessel wall, whereas the majority of pulmonary thrombi had cancer cell clusters, consistent with embolization via blood flow. These cancer cells were immunohistochemically positive for TF (tissue factors) or podoplanin in up to 88% of VTE cases. The frequency of TF-positive monocyte/macrophages in thrombi was higher in cancer-associated VTE than that in VTE without cancer. Citrullinated histone H3 was predominantly observed in the early stages of organizing thrombi. There was no significant difference in thrombus components between VTE with cancer and without cancer groups.
Conclusions: Vascular wall invasion or cancer cell clusters in thrombi might influence thrombogenesis of cancer-associated VTE. TF and podoplanin in cancer cells and in monocyte/macrophages may induce coagulation reactions and platelet aggregation. Neutrophil extracellular traps may play a role in the early stages of VTE, regardless of cancer status.
Keywords: carcinoma; extracellular traps; fibrin; pathology; platelet aggregation; tissue factor; venous thromboembolism.
Figures
Comment in
-
Circulating Tumor Cells and Cancer-Associated Venous Thrombosis: A Missing Link.Arterioscler Thromb Vasc Biol. 2023 Jan;43(1):160-162. doi: 10.1161/ATVBAHA.122.318715. Epub 2022 Dec 1. Arterioscler Thromb Vasc Biol. 2023. PMID: 36453278 Free PMC article. No abstract available.
References
-
- Zamorano JL, Lancellotti P, Rodriguez Munoz D, Aboyans V, Asteggiano R, Galderisi M, Habib G, Lenihan DJ, Lip GYH, Lyon AR, et al. ; ESC Scientific Document Group. 2016 ESC Position Paper on cancer treatments and cardiovascular toxicity developed under the auspices of the ESC Committee for Practice Guidelines: The Task Force for cancer treatments and cardiovascular toxicity of the European Society of Cardiology (ESC). Eur Heart J. 2016;37:2768–2801. doi: 10.1093/eurheartj/ehw211 - PubMed
-
- Timp JF, Braekkan SK, Versteeg HH, Cannegieter SC. Epidemiology of cancer-associated venous thrombosis. Blood. 2013;122:1712–1723. doi: 10.1182/blood-2013-04-460121 - PubMed
-
- Moik F, Prager G, Thaler J, Posch F, Wiedemann S, Schramm T, Englisch C, Mackman N, Pabinger I, Ay C. Hemostatic biomarkers and venous thromboembolism are associated with mortality and response to chemotherapy in patients with pancreatic cancer. Arterioscler Thromb Vasc Biol. 2021;41:2837–2847. doi: 10.1161/ATVBAHA.121.316463 - PubMed
-
- Ogren M, Bergqvist D, Wahlander K, Eriksson H, Sternby NH. Trousseau’s syndrome - what is the evidence? A population-based autopsy study. Thromb Haemost. 2006;95:541–545. doi: 10.1160/TH05-10-0694 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

