Lesions causing post-stroke spasticity localize to a common brain network
- PMID: 36389077
- PMCID: PMC9642815
- DOI: 10.3389/fnagi.2022.1011812
Lesions causing post-stroke spasticity localize to a common brain network
Abstract
Objective: The efficacy of clinical interventions for post-stroke spasticity (PSS) has been consistently unsatisfactory, probably because lesions causing PSS may occur at different locations in the brain, leaving the neuroanatomical substrates of spasticity unclear. Here, we investigated whether heterogeneous lesions causing PSS were localized to a common brain network and then identified the key nodes in this network.
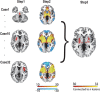
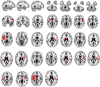
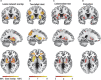
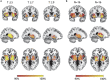
Methods: We used 32 cases of PSS and the Human Connectome dataset (n = 1,000), using a lesion network mapping method to identify the brain regions that were associated with each lesion in patients with PSS. Functional connectivity maps of all lesions were overlaid to identify common connectivity. Furthermore, a split-half replication method was used to evaluate reproducibility. Then, the lesion network mapping results were compared with those of patients with post-stroke non-spastic motor dysfunction (n = 29) to assess the specificity. Next, both sensitive and specific regions associated with PSS were identified using conjunction analyses, and the correlation between these regions and PSS was further explored by correlation analysis.
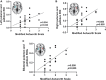
Results: The lesions in all patients with PSS were located in different cortical and subcortical locations. However, at least 93% of these lesions (29/32) had functional connectivity with the bilateral putamen and globus pallidus. These connections were highly repeatable and specific, as compared to those in non-spastic patients. In addition, the functional connectivity between lesions and bilateral putamen and globus pallidus in patients with PSS was positively correlated with the degree of spasticity.
Conclusion: We identified that lesions causing PSS were localized to a common functional connectivity network defined by connectivity to the bilateral putamen and globus pallidus. This network may best cover the locations of lesions causing PSS. The putamen and globus pallidus may be potential key regions in PSS. Our findings complement previous neuroimaging studies on PSS, contributing to identifying patients with stroke at high risk for spasticity at an early stage, and may point to PSS-specific brain stimulation targets.
Keywords: functional connectivity; lesion network mapping; lesions; post-stroke spasticity; resting-state networks.
Copyright © 2022 Qin, Qiu, Liu, Xu, Wang, Guo, Tang and Li.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
LinkOut - more resources
Full Text Sources

