rAAV immunogenicity, toxicity, and durability in 255 clinical trials: A meta-analysis
- PMID: 36389770
- PMCID: PMC9647052
- DOI: 10.3389/fimmu.2022.1001263
rAAV immunogenicity, toxicity, and durability in 255 clinical trials: A meta-analysis
Erratum in
-
Corrigendum: rAAV immunogenicity, toxicity, and durability in 255 clinical trials: A meta-analysis.Front Immunol. 2023 Jan 18;13:1104646. doi: 10.3389/fimmu.2022.1104646. eCollection 2022. Front Immunol. 2023. PMID: 36741359 Free PMC article.
Abstract
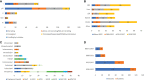
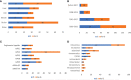
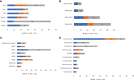
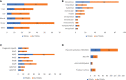
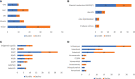
Recombinant Adeno-associated virus (rAAV) is one of the main delivery vectors for gene therapy. To assess immunogenicity, toxicity, and features of AAV gene therapy in clinical settings, a meta-analysis of 255 clinical trials was performed. A total of 7,289 patients are planned to be dosed. AAV2 was the most dominantly used serotype (29.8%, n=72), and 8.3% (n=20) of trials used engineered capsids. 38.7% (n=91) of trials employed neutralizing antibody assays for patient enrollment, while 15.3% (n=36) used ELISA-based total antibody assays. However, there was high variability in the eligibility criteria with cut-off tiers ranging from 1:1 to 1:1,600. To address potential immunogenicity, 46.3% (n=118) of trials applied immunosuppressants (prophylactic or reactive), while 32.7% (n=18) of CNS and 37.5% (n=24) of ocular-directed trials employed immunosuppressants, possibly due to the immune-privileged status of CNS and retina. There were a total of 11 patient deaths across 8 trials, and 18 out of 30 clinical holds were due to toxicity findings in clinical studies. 30.6% (n=78) of trials had treatment-emergent serious adverse events (TESAEs), with hepatotoxicity and thrombotic microangiopathy (systemic delivery) and neurotoxicity (CNS delivery) being the most prominent. Additionally, the durability of gene therapy may be impacted by two distinct decline mechanisms: 1) rapid decline presumably due to immune responses; or 2) gradual decline due to vector dilution. The durability varied significantly depending on disease indication, dose, serotypes, and patient individuals. Most CNS (90.0%) and muscle trials (73.3%) achieved durable transgene expression, while only 43.6% of ocular trials had sustained clinical outcomes. The rAAV production system can affect rAAV quality and thus immunogenicity and toxicity. Out of 186 trials that have disclosed production system information, 63.0% (n=126) of trials used the transient transfection of the HEK293/HEK293T system, while 18.0% (n=36) applied the baculovirus/Sf9 (rBac/Sf9) system. There were no significant differences in TESAEs and durability between AAV generated by rBac/Sf9 and HEK293/HEK293T systems. In summary, rAAV immunogenicity and toxicity poses significant challenges for clinical development of rAAV gene therapies, and it warrants collaborative efforts to standardize monitoring/measurement methods, design novel strategies to overcome immune responses, and openly share relevant information.
Keywords: capsids; clinical trials; gene therapy; immunogenicity; immunosuppressants; neutralizing antibodies; rAAV; toxicity.
Copyright © 2022 Shen, Liu and Ou.
Conflict of interest statement
LO is an employee of Genemagic Biosciences, WS is an employee of Obio Technologies, and SL is an employee of Avirmax.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

