Inferring a spatial code of cell-cell interactions across a whole animal body
- PMID: 36395331
- PMCID: PMC9714814
- DOI: 10.1371/journal.pcbi.1010715
Inferring a spatial code of cell-cell interactions across a whole animal body
Abstract
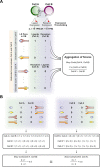
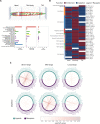
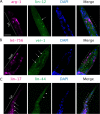
Cell-cell interactions shape cellular function and ultimately organismal phenotype. Interacting cells can sense their mutual distance using combinations of ligand-receptor pairs, suggesting the existence of a spatial code, i.e., signals encoding spatial properties of cellular organization. However, this code driving and sustaining the spatial organization of cells remains to be elucidated. Here we present a computational framework to infer the spatial code underlying cell-cell interactions from the transcriptomes of the cell types across the whole body of a multicellular organism. As core of this framework, we introduce our tool cell2cell, which uses the coexpression of ligand-receptor pairs to compute the potential for intercellular interactions, and we test it across the Caenorhabditis elegans' body. Leveraging a 3D atlas of C. elegans' cells, we also implement a genetic algorithm to identify the ligand-receptor pairs most informative of the spatial organization of cells across the whole body. Validating the spatial code extracted with this strategy, the resulting intercellular distances are negatively correlated with the inferred cell-cell interactions. Furthermore, for selected cell-cell and ligand-receptor pairs, we experimentally confirm the communicatory behavior inferred with cell2cell and the genetic algorithm. Thus, our framework helps identify a code that predicts the spatial organization of cells across a whole-animal body.
Copyright: © 2022 Armingol et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

