Transplantable human thyroid organoids generated from embryonic stem cells to rescue hypothyroidism
- PMID: 36396935
- PMCID: PMC9672394
- DOI: 10.1038/s41467-022-34776-7
Transplantable human thyroid organoids generated from embryonic stem cells to rescue hypothyroidism
Abstract
The thyroid gland captures iodide in order to synthesize hormones that act on almost all tissues and are essential for normal growth and metabolism. Low plasma levels of thyroid hormones lead to hypothyroidism, which is one of the most common disorder in humans and is not always satisfactorily treated by lifelong hormone replacement. Therefore, in addition to the lack of in vitro tractable models to study human thyroid development, differentiation and maturation, functional human thyroid organoids could pave the way to explore new therapeutic approaches. Here we report the generation of transplantable thyroid organoids derived from human embryonic stem cells capable of restoring plasma thyroid hormone in athyreotic mice as a proof of concept for future therapeutic development.
© 2022. The Author(s).
Conflict of interest statement
S.C. and M.R. have filed EU patent regarding the hESC-thyroid derived organoids. All other authors declare no competing interests.
Figures
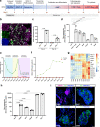
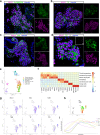
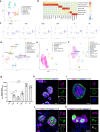
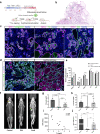
References
-
- Kraut, E. & Farahani, P. A systematic review of clinical practice guidelines’ recommendations on levothyroxine therapy alone versus combination therapy (LT4 plus LT3) for hypothyroidism. Clin. Investigative Med.38, E305-E313 (2015). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

