The endocannabinoid anandamide is an airway relaxant in health and disease
- PMID: 36396957
- PMCID: PMC9672354
- DOI: 10.1038/s41467-022-34327-0
The endocannabinoid anandamide is an airway relaxant in health and disease
Abstract
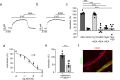
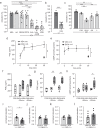
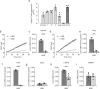
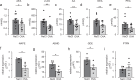
Chronic obstructive airway diseases are a global medical burden that is expected to increase in the near future. However, the underlying mechanistic processes are poorly understood so far. Herein, we show that the endocannabinoid anandamide (AEA) induces prominent airway relaxation in vitro and in vivo. In contrast to 2-arachidonlyglycerol-induced airway relaxation, this is mediated by fatty acid amide hydrolase (FAAH)-dependent metabolites. In particular, we identify mouse and also human epithelial and airway smooth muscle cells as source of AEA-induced prostaglandin E2 production and cAMP as direct mediator of AEA-dependent airway relaxation. Mass spectrometry experiments demonstrate reduced levels of endocannabinoid-like compounds in lungs of ovalbumin-sensitized mice indicating a pathophysiological relevance of endocannabinoid signalling in obstructive airway disease. Importantly, AEA inhalation protects against airway hyper-reactivity after ovalbumin sensitization. Thus, this work highlights the AEA/FAAH axis as a critical regulator of airway tone that could provide therapeutic targets for airway relaxation.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
Endocannabinoid anandamide mediates hypoxic pulmonary vasoconstriction.Proc Natl Acad Sci U S A. 2013 Nov 12;110(46):18710-5. doi: 10.1073/pnas.1308130110. Epub 2013 Oct 28. Proc Natl Acad Sci U S A. 2013. PMID: 24167249 Free PMC article.
-
Role of FAAH-like anandamide transporter in anandamide inactivation.PLoS One. 2013 Nov 4;8(11):e79355. doi: 10.1371/journal.pone.0079355. eCollection 2013. PLoS One. 2013. PMID: 24223930 Free PMC article.
-
Role for fatty acid amide hydrolase (FAAH) in the leptin-mediated effects on feeding and energy balance.Proc Natl Acad Sci U S A. 2018 Jul 17;115(29):7605-7610. doi: 10.1073/pnas.1802251115. Epub 2018 Jul 2. Proc Natl Acad Sci U S A. 2018. PMID: 29967158 Free PMC article.
-
Anandamide and its metabolites: what are their roles in the kidney?Front Biosci (Schol Ed). 2016 Jun 1;8(2):264-77. doi: 10.2741/s461. Front Biosci (Schol Ed). 2016. PMID: 27100705 Free PMC article. Review.
-
Anandamide uptake explained?Trends Pharmacol Sci. 2012 Apr;33(4):181-5. doi: 10.1016/j.tips.2012.01.001. Epub 2012 Jan 31. Trends Pharmacol Sci. 2012. PMID: 22297258 Review.
Cited by
-
Site-specific genetic and functional signatures of aortic endothelial cells at aneurysm predilection sites in healthy and AngII ApoE-/- mice.Angiogenesis. 2024 Nov;27(4):719-738. doi: 10.1007/s10456-024-09933-9. Epub 2024 Jul 4. Angiogenesis. 2024. PMID: 38965173 Free PMC article.
-
New insights into the mechanisms of anandamide-induced airway dilation placing its degradation enzyme, FAAH center stage. Commentary on: Simon A, von Einem T, Seidinger A, Matthey M, Bindila L, Wenzel D (2022) The endocannabinoid anandamide is an airway relaxant in health and disease. Nat Commun 13:6941.Pflugers Arch. 2023 May;475(5):557-559. doi: 10.1007/s00424-023-02802-2. Epub 2023 Mar 1. Pflugers Arch. 2023. PMID: 36856931 Free PMC article. No abstract available.
-
Subnanomolar MAS-related G protein-coupled receptor-X2/B2 antagonists with efficacy in human mast cells and disease models.Signal Transduct Target Ther. 2025 Apr 21;10(1):128. doi: 10.1038/s41392-025-02209-8. Signal Transduct Target Ther. 2025. PMID: 40254631 Free PMC article.
-
Anti-Bacterial and Anti-Biofilm Activities of Anandamide against the Cariogenic Streptococcus mutans.Int J Mol Sci. 2023 Mar 24;24(7):6177. doi: 10.3390/ijms24076177. Int J Mol Sci. 2023. PMID: 37047147 Free PMC article.
References
-
- Wahn H, Wolf J, Kram F, Frantz S, Wagner JA. The endocannabinoid arachidonyl ethanolamide (anandamide) increases pulmonary arterial pressure via cyclooxygenase-2 products in isolated rabbit lungs. Am. J. Physiol. Heart Circ. Physiol. 2005;289:H2491–H2496. doi: 10.1152/ajpheart.00718.2005. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

