Fibrous Cap Thickness Predicts Stable Coronary Plaque Progression: Early Clinical Validation of a Semiautomated OCT Technology
- PMID: 36397766
- PMCID: PMC9668070
- DOI: 10.1016/j.jscai.2022.100400
Fibrous Cap Thickness Predicts Stable Coronary Plaque Progression: Early Clinical Validation of a Semiautomated OCT Technology
Abstract
Background: Imaging-based characteristics associated with the progression of stable coronary atherosclerotic lesions are poorly defined. Utilizing a combination of optical coherence tomography (OCT) and intravascular ultrasound (IVUS) imaging, we aimed to characterize the lesions prone to progression through clinical validation of a semiautomated OCT computational program.
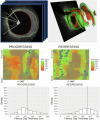
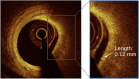
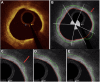
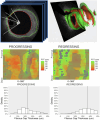
Methods: Patients with stable coronary artery disease underwent nonculprit vessel imaging with IVUS and OCT at baseline and IVUS at the 12-month follow-up. After coregistration of baseline and follow-up IVUS images, paired 5-mm segments from each patient were identified, demonstrating the greatest plaque progression and regression as measured by the change in plaque burden. Experienced readers identified plaque features on corresponding baseline OCT segments, and predictors of plaque progression were assessed by multivariable analysis. Each segment then underwent volumetric assessment of the fibrous cap (FC) using proprietary software.
Results: Among 23 patients (70% men; median age, 67 years), experienced-reader analysis demonstrated that for every 100 μm increase in mean FC thickness, plaques were 87% less likely to progress (P = .01), which persisted on multivariable analysis controlling for baseline plaque burden (P = .05). Automated FC analysis (n = 17 paired segments) confirmed this finding (P = .01) and found thinner minimal FC thickness (P = .01) and larger FC surface area of <65 μm (P = .02) and <100 μm (P = .04) in progressing segments than in regressing segments. No additional imaging features predicted plaque progression.
Conclusions: A semiautomated FC analysis tool confirmed the significant association between thinner FC and stable coronary plaque progression along entire vessel segments, illustrating the diffuse nature of FC thinning and suggesting a future clinical role in predicting the progression of stable coronary artery disease.
Keywords: coronary artery disease; intravascular ultrasound; optical coherence tomography.
Conflict of interest statement
Declaration of competing interest Dr Lopez is a consultant for Abbott-St Jude, runs educational training programs related to optical coherence tomography, and is an investigator in clinical trials with optical coherence tomography. Drs Kassis, Kovarnik, Chen, Weber, Martin, Darki, Woo, Wahle, and Sonka reported no financial interests.
Figures

Similar articles
-
Comparison of optical coherence tomography and intravascular ultrasound for evaluation of coronary lipid-rich atherosclerotic plaque progression and regression.Eur Heart J Cardiovasc Imaging. 2015 Dec;16(12):1374-80. doi: 10.1093/ehjci/jev104. Epub 2015 Apr 24. Eur Heart J Cardiovasc Imaging. 2015. PMID: 25911116
-
Impact of statin therapy on plaque characteristics as assessed by serial OCT, grayscale and integrated backscatter-IVUS.JACC Cardiovasc Imaging. 2012 Feb;5(2):169-77. doi: 10.1016/j.jcmg.2011.11.012. JACC Cardiovasc Imaging. 2012. PMID: 22340823
-
Relationship between arterial and fibrous cap remodeling: a serial three-vessel intravascular ultrasound and optical coherence tomography study.Circ Cardiovasc Interv. 2010 Oct;3(5):484-90. doi: 10.1161/CIRCINTERVENTIONS.109.928911. Epub 2010 Aug 24. Circ Cardiovasc Interv. 2010. PMID: 20736445
-
The diagnostic value of intracoronary optical coherence tomography.Herz. 2011 Aug;36(5):417-29. doi: 10.1007/s00059-011-3487-7. Herz. 2011. PMID: 21744151 Review.
-
Automatic assessment of atherosclerotic plaque features by intracoronary imaging: a scoping review.Front Cardiovasc Med. 2024 Apr 29;11:1332925. doi: 10.3389/fcvm.2024.1332925. eCollection 2024. Front Cardiovasc Med. 2024. PMID: 38742173 Free PMC article.
Cited by
-
Ultrasonic Imaging of Cardiovascular Disease Based on Image Processor Analysis of Hard Plaque Characteristics.Biomed Res Int. 2022 Oct 13;2022:4304524. doi: 10.1155/2022/4304524. eCollection 2022. Biomed Res Int. 2022. Retraction in: Biomed Res Int. 2023 Nov 29;2023:9805045. doi: 10.1155/2023/9805045. PMID: 36277887 Free PMC article. Retracted.
-
Intracoronary Optical Coherence Tomography: Technological Innovations and Clinical Implications in Cardiology.Curr Treat Options Cardiovasc Med. 2025;27(1):44. doi: 10.1007/s11936-025-01103-4. Epub 2025 Jul 24. Curr Treat Options Cardiovasc Med. 2025. PMID: 40727314 Free PMC article. Review.
References
-
- Nicholls S.J., Hsu A., Wolski K., et al. Intravascular ultrasound-derived measures of coronary atherosclerotic plaque burden and clinical outcome. J Am Coll Cardiol. 2010;55(21):2399–2407. - PubMed
-
- Stone P.H., Saito S., Takahashi S., et al. Prediction of progression of coronary artery disease and clinical outcomes using vascular profiling of endothelial shear stress and arterial plaque characteristics: the PREDICTION study. Circulation. 2012;126(2):172–181. - PubMed
-
- Stone G.W., Maehara A., Lansky A.J., et al. A prospective natural-history study of coronary atherosclerosis. N Engl J Med. 2011;364(3):226–235. - PubMed
-
- Tian J., Ren X., Vergallo R., et al. Distinct morphological features of ruptured culprit plaque for acute coronary events compared to those with silent rupture and thin-cap fibroatheroma: a combined optical coherence tomography and intravascular ultrasound study. J Am Coll Cardiol. 2014;63(21):2209–2216. - PubMed
-
- Nissen S.E., Yock P. Intravascular ultrasound: novel pathophysiological insights and current clinical applications. Circulation. 2001;103(4):604–616. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

