Creation of Neuronal Ensembles and Cell-Specific Homeostatic Plasticity through Chronic Sparse Optogenetic Stimulation
- PMID: 36400529
- PMCID: PMC9838708
- DOI: 10.1523/JNEUROSCI.1104-22.2022
Creation of Neuronal Ensembles and Cell-Specific Homeostatic Plasticity through Chronic Sparse Optogenetic Stimulation
Abstract
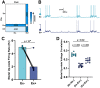
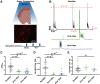
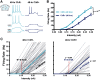
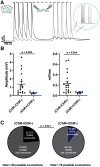
Cortical computations emerge from the dynamics of neurons embedded in complex cortical circuits. Within these circuits, neuronal ensembles, which represent subnetworks with shared functional connectivity, emerge in an experience-dependent manner. Here we induced ensembles in ex vivo cortical circuits from mice of either sex by differentially activating subpopulations through chronic optogenetic stimulation. We observed a decrease in voltage correlation, and importantly a synaptic decoupling between the stimulated and nonstimulated populations. We also observed a decrease in firing rate during Up-states in the stimulated population. These ensemble-specific changes were accompanied by decreases in intrinsic excitability in the stimulated population, and a decrease in connectivity between stimulated and nonstimulated pyramidal neurons. By incorporating the empirically observed changes in intrinsic excitability and connectivity into a spiking neural network model, we were able to demonstrate that changes in both intrinsic excitability and connectivity accounted for the decreased firing rate, but only changes in connectivity accounted for the observed decorrelation. Our findings help ascertain the mechanisms underlying the ability of chronic patterned stimulation to create ensembles within cortical circuits and, importantly, show that while Up-states are a global network-wide phenomenon, functionally distinct ensembles can preserve their identity during Up-states through differential firing rates and correlations.SIGNIFICANCE STATEMENT The connectivity and activity patterns of local cortical circuits are shaped by experience. This experience-dependent reorganization of cortical circuits is driven by complex interactions between different local learning rules, external input, and reciprocal feedback between many distinct brain areas. Here we used an ex vivo approach to demonstrate how simple forms of chronic external stimulation can shape local cortical circuits in terms of their correlated activity and functional connectivity. The absence of feedback between different brain areas and full control of external input allowed for a tractable system to study the underlying mechanisms and development of a computational model. Results show that differential stimulation of subpopulations of neurons significantly reshapes cortical circuits and forms subnetworks referred to as neuronal ensembles.
Keywords: Up-states; computational model; homeostatic plasticity; neural dynamics; neuronal ensembles.
Copyright © 2023 the authors.
Figures


Similar articles
-
Intrinsic excitability mechanisms of neuronal ensemble formation.Elife. 2022 May 4;11:e77470. doi: 10.7554/eLife.77470. Elife. 2022. PMID: 35506662 Free PMC article.
-
Rapid Disinhibition by Adjustment of PV Intrinsic Excitability during Whisker Map Plasticity in Mouse S1.J Neurosci. 2018 May 16;38(20):4749-4761. doi: 10.1523/JNEUROSCI.3628-17.2018. Epub 2018 Apr 20. J Neurosci. 2018. PMID: 29678876 Free PMC article.
-
M-Current Inhibition in Hippocampal Excitatory Neurons Triggers Intrinsic and Synaptic Homeostatic Responses at Different Temporal Scales.J Neurosci. 2020 May 6;40(19):3694-3706. doi: 10.1523/JNEUROSCI.1914-19.2020. Epub 2020 Apr 10. J Neurosci. 2020. PMID: 32277041 Free PMC article.
-
Homeostatic plasticity in neural development.Neural Dev. 2018 Jun 1;13(1):9. doi: 10.1186/s13064-018-0105-x. Neural Dev. 2018. PMID: 29855353 Free PMC article. Review.
-
Network homeostasis: a matter of coordination.Curr Opin Neurobiol. 2009 Apr;19(2):168-73. doi: 10.1016/j.conb.2009.05.012. Epub 2009 Jun 18. Curr Opin Neurobiol. 2009. PMID: 19540746 Free PMC article. Review.
Cited by
-
Oxytocin Modifies the Excitability and the Action Potential Shape of the Hippocampal CA1 GABAergic Interneurons.Int J Mol Sci. 2024 Feb 23;25(5):2613. doi: 10.3390/ijms25052613. Int J Mol Sci. 2024. PMID: 38473860 Free PMC article.
-
Revealing and reshaping attractor dynamics in large networks of cortical neurons.PLoS Comput Biol. 2024 Jan 19;20(1):e1011784. doi: 10.1371/journal.pcbi.1011784. eCollection 2024 Jan. PLoS Comput Biol. 2024. PMID: 38241417 Free PMC article.
-
Optogenetic stimulation shapes dendritic trees of infragranular cortical pyramidal cells.Front Cell Neurosci. 2023 Aug 1;17:1212483. doi: 10.3389/fncel.2023.1212483. eCollection 2023. Front Cell Neurosci. 2023. PMID: 37587917 Free PMC article.
-
Microstimulation of sensory cortex engages natural sensory representations.Curr Biol. 2023 May 8;33(9):1765-1777.e5. doi: 10.1016/j.cub.2023.03.085. Epub 2023 May 2. Curr Biol. 2023. PMID: 37130521 Free PMC article.
-
Ex vivo cortical circuits learn to predict and spontaneously replay temporal patterns.Nat Commun. 2025 Apr 4;16(1):3179. doi: 10.1038/s41467-025-58013-z. Nat Commun. 2025. PMID: 40185714 Free PMC article.
