Engineering white blood cell membrane-camouflaged nanocarriers for inflammation-related therapeutics
- PMID: 36406250
- PMCID: PMC9650012
- DOI: 10.1016/j.bioactmat.2022.10.026
Engineering white blood cell membrane-camouflaged nanocarriers for inflammation-related therapeutics
Abstract
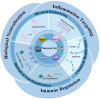
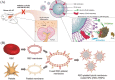
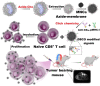
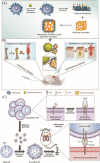
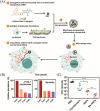
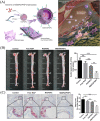
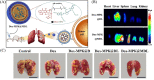
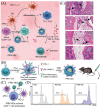
White blood cells (WBCs) play essential roles against inflammatory disorders, bacterial infections, and cancers. Inspired by nature, WBC membrane-camouflaged nanocarriers (WBC-NCs) have been developed to mimic the "dynamic" functions of WBCs, such as transendothelial migration, adhesion to injured blood vessels, etc, which make them promising for diverse medical applications. WBC-NCs inherit the cell membrane antigens of WBCs, while still exhibiting the robust inflammation-related therapeutic potential of synthetic nanocarriers with excellent (bio)physicochemical performance. This review summarizes the proposed concept of cell membrane engineering, which utilizes physical engineering, chemical modification, and biological functionalization technologies to endow the natural cell membrane with abundant functionalities. In addition, it highlights the recent progress and applications of WBC-NCs for inflammation targeting, biological neutralization, and immune modulation. Finally, the challenges and opportunities in realizing the full potential of WBC-NCs for the manipulation of inflammation-related therapeutics are discussed.
Keywords: Biological neutralization; Immune modulation; Inflammation targeting; White blood cell membrane-camouflaged nanocarriers.
© 2022 The Authors.
Conflict of interest statement
All authors have given approval to the final version of the manuscript. The authors declare no competing financial interest.
Figures






Similar articles
-
White Blood Cell Membrane-Coated Nanoparticles: Recent Development and Medical Applications.Adv Healthc Mater. 2022 Apr;11(7):e2101349. doi: 10.1002/adhm.202101349. Epub 2021 Aug 31. Adv Healthc Mater. 2022. PMID: 34468090 Review.
-
Engineered Cell Membrane-Derived Nanocarriers: The Enhanced Delivery System for Therapeutic Applications.Front Cell Dev Biol. 2022 Feb 28;10:844050. doi: 10.3389/fcell.2022.844050. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 35295856 Free PMC article. Review.
-
Cell Membrane-Camouflaged Nanocarriers with Biomimetic Deformability of Erythrocytes for Ultralong Circulation and Enhanced Cancer Therapy.ACS Nano. 2022 Apr 26;16(4):6527-6540. doi: 10.1021/acsnano.2c00893. Epub 2022 Apr 15. ACS Nano. 2022. PMID: 35426300
-
Hybrid cell membrane-coated nanoparticles: A multifunctional biomimetic platform for cancer diagnosis and therapy.Acta Biomater. 2020 Aug;112:1-13. doi: 10.1016/j.actbio.2020.05.028. Epub 2020 May 26. Acta Biomater. 2020. PMID: 32470527 Review.
-
Cell Membrane-Camouflaged Nanocarriers for Cancer Diagnostic and Therapeutic.Front Pharmacol. 2020 Feb 4;11:24. doi: 10.3389/fphar.2020.00024. eCollection 2020. Front Pharmacol. 2020. PMID: 32116701 Free PMC article. Review.
Cited by
-
Neutrophil-like pH-responsive pro-efferocytic nanoparticles improve neurological recovery by promoting erythrophagocytosis after intracerebral hemorrhage.Theranostics. 2024 Jan 1;14(1):283-303. doi: 10.7150/thno.90370. eCollection 2024. Theranostics. 2024. PMID: 38164152 Free PMC article.
-
Epigallocatechin-3-Gallate attenuates lipopolysacharide-induced pneumonia via modification of inflammation, oxidative stress, apoptosis, and autophagy.BMC Complement Med Ther. 2024 Apr 5;24(1):147. doi: 10.1186/s12906-024-04436-y. BMC Complement Med Ther. 2024. PMID: 38580929 Free PMC article.
-
Biomimetic nanovaccines in cancer therapy: mechanisms, efficacy, and clinical translation.Mater Today Bio. 2025 Jul 18;34:102116. doi: 10.1016/j.mtbio.2025.102116. eCollection 2025 Oct. Mater Today Bio. 2025. PMID: 40791791 Free PMC article. Review.
-
Engineered Cell Membrane-Camouflaged Nanomaterials for Biomedical Applications.Nanomaterials (Basel). 2024 Feb 23;14(5):413. doi: 10.3390/nano14050413. Nanomaterials (Basel). 2024. PMID: 38470744 Free PMC article. Review.
-
Nature-inspired nanocarriers for improving drug therapy of atherosclerosis.Regen Biomater. 2023 Aug 11;10:rbad069. doi: 10.1093/rb/rbad069. eCollection 2023. Regen Biomater. 2023. PMID: 37641591 Free PMC article. Review.
References
-
- Netea M.G., Balkwill F., Chonchol M., Cominelli F., Donath M.Y., Giamarellos-Bourboulis E.J., Golenbock D., Gresnigt M.S., Heneka M.T., Hoffman H.M., Hotchkiss R., Joosten L.A.B., Kastner D.L., Korte M., Latz E., Libby P., Mandrup-Poulsen T., Mantovani A., Mills K.H.G., Nowak K.L., O'Neill L.A., Pickkers P., van der Poll T., Ridker P.M., Schalkwijk J., Schwartz D.A., Siegmund B., Steer C.J., Tilg H., van der Meer J.W.M., van de Veerdonk F.L., Dinarello C.A. A guiding map for inflammation. Nat. Immunol. 2017;18:826–831. doi: 10.1038/ni.3790. - DOI - PMC - PubMed
-
- Shankar-Hari M., Phillips G.S., Levy M.L., Seymour C.W., Liu V.X., Deutschman C.S., Angus D.C., Rubenfeld G.D., Singer M., for the S.D.T. Force Developing a new definition and assessing new clinical criteria for septic shock: for the third international consensus definitions for sepsis and septic shock (Sepsis-3) JAMA. 2016;315:775–787. doi: 10.1001/jama.2016.0289. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

