Dihydropyrimidine Dehydrogenase-Mediated Resistance to 5-Fluorouracil: Mechanistic Investigation and Solution
- PMID: 36407958
- PMCID: PMC9667542
- DOI: 10.1021/acsptsci.2c00117
Dihydropyrimidine Dehydrogenase-Mediated Resistance to 5-Fluorouracil: Mechanistic Investigation and Solution
Abstract
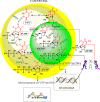
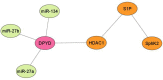
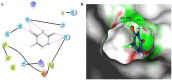
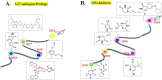
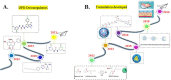
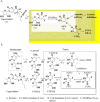
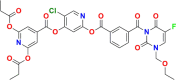
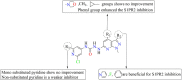
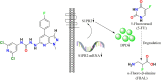
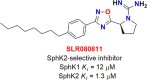
5-Fluorouracil (5-FU) is one of the most widely used chemotherapeutics for the treatment of cancers associated with the aerodigestive tract, breast, and colorectal system. The efficacy of 5-FU is majorly affected by dihydropyrimidine dehydrogenase (DPD) as it degrades more than 80% of administered 5-FU into an inactive metabolite, dihydrofluorouracil. Herein we discuss the molecular mechanism of this inactivation by analyzing the interaction pattern and electrostatic complementarity of the DPD-5-FU complex. The basis of DPD overexpression in cancer cell lines due to significantly distinct levels of the miRNAs (miR-134, miR-27b, and miR-27a) compared to normal cells has also been outlined. Additionally, some kinases including sphingosine kinase 2 (SphK2) have been reported to correlate with DPD expression. Currently, to address this problem various strategies are reported in the literature, including 5-FU analogues (bypass the DPD-mediated inactivation), DPD downregulators (regulate the DPD expression levels in tumors), inhibitors (as promising adjuvants), and formulation development loaded with 5-FU (liposomes, nanoparticles, nanogels, etc.), which are briefly discussed in this Review.
© 2022 American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
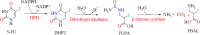



Similar articles
-
microRNAs miR-27a and miR-27b directly regulate liver dihydropyrimidine dehydrogenase expression through two conserved binding sites.Mol Cancer Ther. 2014 Mar;13(3):742-51. doi: 10.1158/1535-7163.MCT-13-0878. Epub 2014 Jan 8. Mol Cancer Ther. 2014. PMID: 24401318 Free PMC article.
-
Dihydropyrimidine dehydrogenase (DPD) expression is negatively regulated by certain microRNAs in human lung tissues.Lung Cancer. 2012 Jul;77(1):16-23. doi: 10.1016/j.lungcan.2011.12.018. Epub 2012 Feb 3. Lung Cancer. 2012. PMID: 22306127
-
SphK2 confers 5-fluorouracil resistance to colorectal cancer via upregulating H3K56ac-mediated DPD expression.Oncogene. 2020 Jul;39(29):5214-5227. doi: 10.1038/s41388-020-1352-y. Epub 2020 Jun 16. Oncogene. 2020. PMID: 32546724
-
Implications of dihydropyrimidine dehydrogenase on 5-fluorouracil pharmacogenetics and pharmacogenomics.Pharmacogenomics. 2002 Jul;3(4):485-92. doi: 10.1517/14622416.3.4.485. Pharmacogenomics. 2002. PMID: 12164772 Review.
-
[Dihydropyrimidine dehydrogenase activity and its genetic aberrations].Gan To Kagaku Ryoho. 2006 Aug;33(8):1041-8. Gan To Kagaku Ryoho. 2006. PMID: 16912518 Review. Japanese.
Cited by
-
Advanced prodrug strategies in nucleoside analogues targeting the treatment of gastrointestinal malignancies.Front Cell Dev Biol. 2023 Apr 18;11:1173432. doi: 10.3389/fcell.2023.1173432. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 37143892 Free PMC article. Review.
-
Acquired resistance to molecularly targeted therapies for cancer.Cancer Drug Resist. 2025 Jun 5;8:27. doi: 10.20517/cdr.2024.189. eCollection 2025. Cancer Drug Resist. 2025. PMID: 40510029 Free PMC article. Review.
-
Hepatoprotective effect of Nobiletin against 5-fluorouracil induce hepatotoxicity.Curr Res Pharmacol Drug Discov. 2024 Sep 12;7:100199. doi: 10.1016/j.crphar.2024.100199. eCollection 2024. Curr Res Pharmacol Drug Discov. 2024. PMID: 39411523 Free PMC article.
-
Endogenous stimuli-responsive separating microneedles to inhibit hypertrophic scar through remodeling the pathological microenvironment.Nat Commun. 2024 Mar 6;15(1):2038. doi: 10.1038/s41467-024-46328-2. Nat Commun. 2024. PMID: 38448448 Free PMC article.
References
-
- Duschinsky R.; Pleven E.; Heidelberger C. The synthesis of 5-fluoropyrimidines. J. Am. Chem. Soc. 1957, 79 (16), 4559–4560. 10.1021/ja01573a087. - DOI
Publication types
LinkOut - more resources
Full Text Sources
