Long-term effects of prenatal undernutrition on female rat hypothalamic KNDy neurons
- PMID: 36408965
- PMCID: PMC9782422
- DOI: 10.1530/EC-22-0307
Long-term effects of prenatal undernutrition on female rat hypothalamic KNDy neurons
Abstract
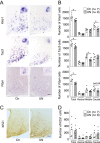
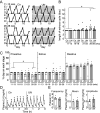
The nutritional environment during development periods induces metabolic programming, leading to metabolic disorders and detrimental influences on human reproductive health. This study aimed to determine the long-term adverse effect of intrauterine malnutrition on the reproductive center kisspeptin-neurokinin B-dynorphin A (KNDy) neurons in the hypothalamic arcuate nucleus (ARC) of female offspring. Twelve pregnant rats were divided into ad-lib-fed (control, n = 6) and 50% undernutrition (UN, n = 6) groups. The UN group was restricted to 50% daily food intake of the control dams from gestation day 9 until term delivery. Differences between the two groups in terms of various maternal parameters, including body weight (BW), pregnancy duration, and litter size, as well as birth weight, puberty onset, estrous cyclicity, pulsatile luteinizing hormone (LH) secretion, and hypothalamic gene expression of offspring, were determined. Female offspring of UN dams exhibited low BW from birth to 3 weeks, whereas UN offspring showed signs of precocious puberty; hypothalamic Tac3 (a neurokinin B gene) expression was increased in prepubertal UN offspring, and the BW at the virginal opening was lower in UN offspring than that in the control group. Interestingly, the UN offspring showed significant decreases in the number of KNDy gene-expressing cells after 29 weeks of age, but the number of ARC kisspeptin-immunoreactive cells, pulsatile LH secretions, and estrous cyclicity were comparable between the groups. In conclusion, intrauterine undernutrition induced various changes in KNDy gene expression depending on the life stage. Thus, intrauterine undernutrition affected hypothalamic developmental programming in female rats.
Keywords: fetal programing; kisspeptin; low birth weight; prenatal undernutrition.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


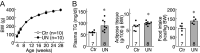
References
LinkOut - more resources
Full Text Sources

