The fate of interneurons, GABAA receptor sub-types and perineuronal nets in Alzheimer's disease
- PMID: 36409151
- PMCID: PMC9836378
- DOI: 10.1111/bpa.13129
The fate of interneurons, GABAA receptor sub-types and perineuronal nets in Alzheimer's disease
Abstract
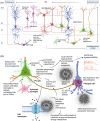
Alzheimer's disease (AD) is the most common neurological disease, which is associated with gradual memory loss and correlated with synaptic hyperactivity and abnormal oscillatory rhythmic brain activity that precedes phenotypic alterations and is partly responsible for the spread of the disease pathology. Synaptic hyperactivity is thought to be because of alteration in the homeostasis of phasic and tonic synaptic inhibition, which is orchestrated by the GABAA inhibitory system, encompassing subclasses of interneurons and GABAA receptors, which play a vital role in cognitive functions, including learning and memory. Furthermore, the extracellular matrix, the perineuronal nets (PNNs) which often go unnoticed in considerations of AD pathology, encapsulate the inhibitory cells and neurites in critical brain regions and have recently come under the light for their crucial role in synaptic stabilisation and excitatory-inhibitory balance and when disrupted, serve as a potential trigger for AD-associated synaptic imbalance. Therefore, in this review, we summarise the current understanding of the selective vulnerability of distinct interneuron subtypes, their synaptic and extrasynaptic GABAA R subtypes as well as the changes in PNNs in AD, detailing their contribution to the mechanisms of disease development. We aim to highlight how seemingly unique malfunction in each component of the interneuronal GABA inhibitory system can be tied together to result in critical circuit dysfunction, leading to the irreversible symptomatic damage observed in AD.
Keywords: Alzheimer's disease; GABA; Synaptic; interneurons; neurodegeneration; perineuronal nets.
© 2022 The Authors. Brain Pathology published by John Wiley & Sons Ltd on behalf of International Society of Neuropathology.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Walters A, Phillips E, Zheng R, Biju M, Kuruvilla T. Evidence for neuroinflammation in Alzheimer's disease. Prog Neurol Psychiatry. 2016;20:25–31.
-
- Busche MA, Eichhoff G, Adelsberger H, Abramowski D, Wiederhold KH, Haass C, et al. Clusters of hyperactive neurons near amyloid plaques in a mouse model of Alzheimer's disease. Science. 2008;321:1686–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

