PD-1-CD28 fusion protein strengthens mesothelin-specific TRuC T cells in preclinical solid tumor models
- PMID: 36409438
- PMCID: PMC9947055
- DOI: 10.1007/s13402-022-00747-9
PD-1-CD28 fusion protein strengthens mesothelin-specific TRuC T cells in preclinical solid tumor models
Abstract
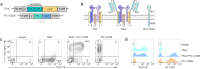
Background: T cell receptor fusion constructs (TRuC) consist of an antibody-based single chain variable fragment (scFv) fused to a T cell receptor chain (TCR) and allow recognition of cancer cells in an HLA-independent manner. Unlike chimeric antigen receptors (CAR), TRuC are integrated into the TCR complex resulting in a functional chimera with novel specificity, whilst retaining TCR signaling. To further enhance anti-tumor function, we expressed a PD-1-CD28 fusion receptor in TRuC T cells aiming to prevent tumor-induced immune suppression and T cell anergy.
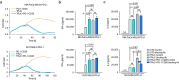
Methods: The activation level of engineered T cells was investigated in co-culture experiments with tumor cells followed by quantification of released cytokines using ELISA. To study T cell-mediated tumor cell lysis in vitro, impedance-based real-time tumor cell killing and LDH release was measured. Finally, two xenograft mouse cancer models were employed to explore the therapeutic potential of engineered T cells.
Results: In co-culture assays, co-expression of PD-1-CD28 enhanced cytokine production of TRuC T cells. This effect was dependent on PD-L1 to PD-1-CD28 interactions, as blockade of PD-L1 amplified IFN-γ production in unmodified TRuC T cells to a greater level compared to TRuC-PD-1-CD28 T cells. In vivo, PD-1-CD28 co-expression supported the anti-tumor efficacy of TRuC T cells in two xenograft mouse cancer models.
Conclusion: Together, these results demonstrate the therapeutic potential of PD-1-CD28 co-expression in TRuC T cells to prevent PD-L1-induced T cell hypofunction.
Keywords: Adoptive cell transfer; Immunosuppression; PD-1-CD28 fusion protein; T cell hypofunction; TRuC T cells.
© 2022. The Author(s).
Conflict of interest statement
S.E. and S.K. received research support from TCR2 Inc for the presented study. S.K. and S.E. have licensed I.P. to TCR2 Inc and have received licensing payments. S.K. has received honoraria from TCR2 Inc, Novartis, BMS and GSK. S.K. has received research support from Arcus Biosciences and Tabby Therapeutics for work unrelated to the present study. S.K. and S.E. have licensed I.P. to Carina Biotech and received licensing payments. The authors declare that no other competing financial interest nor conflict of interest exists.
Figures

Similar articles
-
Functional enhancement of mesothelin-targeted TRuC-T cells by a PD1-CD28 chimeric switch receptor.Cancer Immunol Immunother. 2023 Dec;72(12):4195-4207. doi: 10.1007/s00262-023-03556-7. Epub 2023 Oct 18. Cancer Immunol Immunother. 2023. PMID: 37848682 Free PMC article.
-
CD19-specific CAR T Cells that Express a PD-1/CD28 Chimeric Switch-Receptor are Effective in Patients with PD-L1-positive B-Cell Lymphoma.Clin Cancer Res. 2021 Jan 15;27(2):473-484. doi: 10.1158/1078-0432.CCR-20-1457. Epub 2020 Oct 7. Clin Cancer Res. 2021. PMID: 33028589 Clinical Trial.
-
4-1BB-Based CAR T Cells Effectively Reverse Exhaustion and Enhance the Anti-Tumor Immune Response through Autocrine PD-L1 scFv Antibody.Int J Mol Sci. 2023 Feb 20;24(4):4197. doi: 10.3390/ijms24044197. Int J Mol Sci. 2023. PMID: 36835603 Free PMC article.
-
Roles of PD-1/PD-L1 Pathway: Signaling, Cancer, and Beyond.Adv Exp Med Biol. 2020;1248:33-59. doi: 10.1007/978-981-15-3266-5_3. Adv Exp Med Biol. 2020. PMID: 32185706 Review.
-
Novel strategies for inhibiting PD-1 pathway-mediated immune suppression while simultaneously delivering activating signals to tumor-reactive T cells.Cancer Immunol Immunother. 2015 Oct;64(10):1287-93. doi: 10.1007/s00262-015-1677-5. Epub 2015 Mar 20. Cancer Immunol Immunother. 2015. PMID: 25792524 Free PMC article. Review.
Cited by
-
Functional enhancement of mesothelin-targeted TRuC-T cells by a PD1-CD28 chimeric switch receptor.Cancer Immunol Immunother. 2023 Dec;72(12):4195-4207. doi: 10.1007/s00262-023-03556-7. Epub 2023 Oct 18. Cancer Immunol Immunother. 2023. PMID: 37848682 Free PMC article.
-
CD28 co-stimulation: novel insights and applications in cancer immunotherapy.Nat Rev Immunol. 2024 Dec;24(12):878-895. doi: 10.1038/s41577-024-01061-1. Epub 2024 Jul 25. Nat Rev Immunol. 2024. PMID: 39054343 Review.
-
From barriers to novel strategies: smarter CAR T therapy hits hard to tumors.Front Immunol. 2023 Jul 14;14:1203230. doi: 10.3389/fimmu.2023.1203230. eCollection 2023. Front Immunol. 2023. PMID: 37520522 Free PMC article. Review.
-
Learning from the microbes: exploiting the microbiome to enforce T cell immunotherapy.Front Immunol. 2023 Sep 18;14:1269015. doi: 10.3389/fimmu.2023.1269015. eCollection 2023. Front Immunol. 2023. PMID: 37799719 Free PMC article. Review.
-
Unlocking the potential of Tregs: innovations in CAR technology.Front Mol Biosci. 2023 Oct 12;10:1267762. doi: 10.3389/fmolb.2023.1267762. eCollection 2023. Front Mol Biosci. 2023. PMID: 37900916 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

