A natural history study to track brain and spinal cord changes in individuals with Friedreich's ataxia: TRACK-FA study protocol
- PMID: 36410013
- PMCID: PMC9678384
- DOI: 10.1371/journal.pone.0269649
A natural history study to track brain and spinal cord changes in individuals with Friedreich's ataxia: TRACK-FA study protocol
Erratum in
-
Correction: A natural history study to track brain and spinal cord changes in individuals with Friedreich's ataxia: TRACK-FA study protocol.PLoS One. 2025 Mar 18;20(3):e0320683. doi: 10.1371/journal.pone.0320683. eCollection 2025. PLoS One. 2025. PMID: 40100780 Free PMC article.
-
Correction: A natural history study to track brain and spinal cord changes in individuals with Friedreich's ataxia: TRACK-FA study protocol.PLoS One. 2025 May 22;20(5):e0324751. doi: 10.1371/journal.pone.0324751. eCollection 2025. PLoS One. 2025. PMID: 40403010 Free PMC article.
Abstract
Introduction: Drug development for neurodegenerative diseases such as Friedreich's ataxia (FRDA) is limited by a lack of validated, sensitive biomarkers of pharmacodynamic response in affected tissue and disease progression. Studies employing neuroimaging measures to track FRDA have thus far been limited by their small sample sizes and limited follow up. TRACK-FA, a longitudinal, multi-site, and multi-modal neuroimaging natural history study, aims to address these shortcomings by enabling better understanding of underlying pathology and identifying sensitive, clinical trial ready, neuroimaging biomarkers for FRDA.
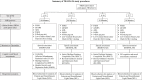
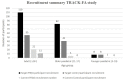
Methods: 200 individuals with FRDA and 104 control participants will be recruited across seven international study sites. Inclusion criteria for participants with genetically confirmed FRDA involves, age of disease onset ≤ 25 years, Friedreich's Ataxia Rating Scale (FARS) functional staging score of ≤ 5, and a total modified FARS (mFARS) score of ≤ 65 upon enrolment. The control cohort is matched to the FRDA cohort for age, sex, handedness, and years of education. Participants will be evaluated at three study visits over two years. Each visit comprises of a harmonized multimodal Magnetic Resonance Imaging (MRI) and Spectroscopy (MRS) scan of the brain and spinal cord; clinical, cognitive, mood and speech assessments and collection of a blood sample. Primary outcome measures, informed by previous neuroimaging studies, include measures of: spinal cord and brain morphometry, spinal cord and brain microstructure (measured using diffusion MRI), brain iron accumulation (using Quantitative Susceptibility Mapping) and spinal cord biochemistry (using MRS). Secondary and exploratory outcome measures include clinical, cognitive assessments and blood biomarkers.
Discussion: Prioritising immediate areas of need, TRACK-FA aims to deliver a set of sensitive, clinical trial-ready neuroimaging biomarkers to accelerate drug discovery efforts and better understand disease trajectory. Once validated, these potential pharmacodynamic biomarkers can be used to measure the efficacy of new therapeutics in forestalling disease progression.
Clinical trial registration: ClinicalTrails.gov Identifier: NCT04349514.
Copyright: © 2022 Georgiou-Karistianis et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
I have read the journal’s policy and the authors of this manuscript have the following competing interests: R.E. and S.Z. are employed by Takeda Pharmaceutical Company Ltd with both authors receiving salary and S.Z. holds stocks in the company. A.S. was employed by Takeda Pharmaceutical Company Ltd at the time of his contribution to the TRACK-FA project. Takeda Pharmaceutical Company Ltd remains committed to FRDA research and will help develop translational tools to monitor patient disease and share with the FRDA community. T.S. and B.Y. are both employees of PTC Therapeutics. D.L. is a grant recipient from the National Institute of Health (NIH), Muscular Dystrophy Association (MDA), Friedreich’s Ataxia Research Alliance (FARA), Reata Pharmaceuticals Inc, Retrotope Inc, Voyager Therapeutics, Novartis Gene Therapies, Audentes Therapeutics (Astellas Gene Therapies) and Minoryx Therapeutics S.L. T.P.L.R. has equity interest in PRISM Clinical Imaging and Proteus Neurodynamics and consulting/advisory board engagement with CTF MEG International Services LP, Ricoh Company Ltd, Spago Nanomedical AB, Avexis (Novartis Gene Therapies) and Acadia Pharmaceuticals Inc. P.G.H. is a grant recipient from the Friedreich’s Ataxia Research Alliance (FARA), GoFAR, Ataxia UK, the Bob Allison Ataxia Research Centre, and the National Institute of Health (NIH). CMRR is supported by NIH grants P41EB027061 and P30NS076408. P.G.H. reports grants from Minoryx Therapeutics for activities outside this study. M.P. and R.J. and M.L. are employed by IXICO plc, ML is a shareholder for IXICO plc. M.C.F. is a grant recipient from PTC Therapeutics and has taken part in advisory board for PTC Therapeutics and Avexis (Novartis Gene Therapies). T.R. is a grant recipient from the Friedreich’s Ataxia Research Alliance (FARA). C.L. is a research grant recipient from the Friedreich’s Ataxia Research Alliance (FARA), GoFAR, Ataxia UK, the Bob Allison Ataxia Research Center, and National Institute of Health (NIH) grants P41. EB027061 and P30 NS076408. C.L. reports research grants from Minoryx Therapeutics and Biogen Inc. for activities outside this study. S.S. is a broad member of the Research Advisory Board for National Ataxia Foundation (USA), a research grant recipient from the Friedreich’s Ataxia Research Alliance (FARA), Wyck Foundation, National Ataxia Foundation, Muscular Dystrophy Association (MDA), National Institute of Health (NIH), FDA and receives industry support from Reata Pharmaceutical Inc, Retrotope Inc, PTC Therapeutics, Biohaven Pharmaceuticals, Avidity Biosciences Inc, and Strides Pharma Science Limited. M.L.K. holds shares in Novartis Gene Therapies. K.R. has received grants from the German Federal Ministry of Education and Research (BMBF 01GQ1402, 01DN18022), the German Research Foundation (IRTG 2150, ZUK32/1), Alzheimer Forschung Initiative e.V. (AFI 13812, NL-18002CB) and honoraria for presentations or advisory boards from Biogen and Roche. J.F. is employed by the Friedreich’s Ataxia Research Alliance (FARA) and receives a salary from this institution. L.C. is a research grant recipient from the Friedreich Ataxia Research Alliance (FARA), Ataxia UK, Medical Research Future Fund and is funded by a Medical Research Futures Fund Next Generation Career Development Fellowship.
Figures
Similar articles
-
Neuroimaging Biomarkers for Friedreich Ataxia: A Cross-Sectional Analysis of the TRACK-FA Study.Ann Neurol. 2025 Aug;98(2):386-397. doi: 10.1002/ana.27237. Epub 2025 Mar 22. Ann Neurol. 2025. PMID: 40119735 Free PMC article.
-
Progressive Spinal Cord Degeneration in Friedreich's Ataxia: Results from ENIGMA-Ataxia.Mov Disord. 2023 Jan;38(1):45-56. doi: 10.1002/mds.29261. Epub 2022 Oct 29. Mov Disord. 2023. PMID: 36308733 Free PMC article.
-
Tract-Specific Spinal Cord Diffusion Tensor Imaging in Friedreich's Ataxia.Mov Disord. 2022 Feb;37(2):354-364. doi: 10.1002/mds.28841. Epub 2021 Oct 29. Mov Disord. 2022. PMID: 34713932
-
Therapeutic Biomarkers in Friedreich's Ataxia: a Systematic Review and Meta-analysis.Cerebellum. 2024 Jun;23(3):1184-1203. doi: 10.1007/s12311-023-01621-6. Epub 2023 Oct 27. Cerebellum. 2024. PMID: 37889470 Free PMC article.
-
Clinical evidence of interventions assessed in Friedreich ataxia: a systematic review.Ther Adv Rare Dis. 2022 Nov 29;3:26330040221139872. doi: 10.1177/26330040221139872. eCollection 2022 Jan-Dec. Ther Adv Rare Dis. 2022. PMID: 37180421 Free PMC article. Review.
Cited by
-
Correction: A natural history study to track brain and spinal cord changes in individuals with Friedreich's ataxia: TRACK-FA study protocol.PLoS One. 2025 May 22;20(5):e0324751. doi: 10.1371/journal.pone.0324751. eCollection 2025. PLoS One. 2025. PMID: 40403010 Free PMC article.
-
Spinal cord magnetic resonance imaging and spectroscopy detect early-stage alterations and disease progression in Friedreich ataxia.Brain Commun. 2022 Oct 3;4(5):fcac246. doi: 10.1093/braincomms/fcac246. eCollection 2022. Brain Commun. 2022. PMID: 36300142 Free PMC article.
-
MR Imaging in Ataxias: Consensus Recommendations by the Ataxia Global Initiative Working Group on MRI Biomarkers.Cerebellum. 2024 Jun;23(3):931-945. doi: 10.1007/s12311-023-01572-y. Epub 2023 Jun 6. Cerebellum. 2024. PMID: 37280482 Free PMC article. Review.
-
New and Emerging Drug and Gene Therapies for Friedreich Ataxia.CNS Drugs. 2024 Oct;38(10):791-805. doi: 10.1007/s40263-024-01113-z. Epub 2024 Aug 8. CNS Drugs. 2024. PMID: 39115603 Free PMC article. Review.
-
The Natural History Study and Biomarker Collection of the Clinical Research Consortium for the Study of Cerebellar Ataxia (CRC-SCA).Cerebellum. 2025 Jul 18;24(5):134. doi: 10.1007/s12311-025-01885-0. Cerebellum. 2025. PMID: 40679685 Review.
References
-
- Reetz K, Dogan I, Hohenfeld C, Didszun C, Giunti P, Mariotti C, et al.. Nonataxia symptoms in Friedreich Ataxia: Report from the Registry of the European Friedreich’s Ataxia Consortium for Translational Studies (EFACTS). Neurology. 2018;91(10):e917–e30. doi: 10.1212/WNL.0000000000006121 - DOI - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

