The Potential Contribution of Hexavalent Chromium to the Carcinogenicity of Chrysotile Asbestos
- PMID: 36410050
- PMCID: PMC9768810
- DOI: 10.1021/acs.chemrestox.2c00314
The Potential Contribution of Hexavalent Chromium to the Carcinogenicity of Chrysotile Asbestos
Abstract
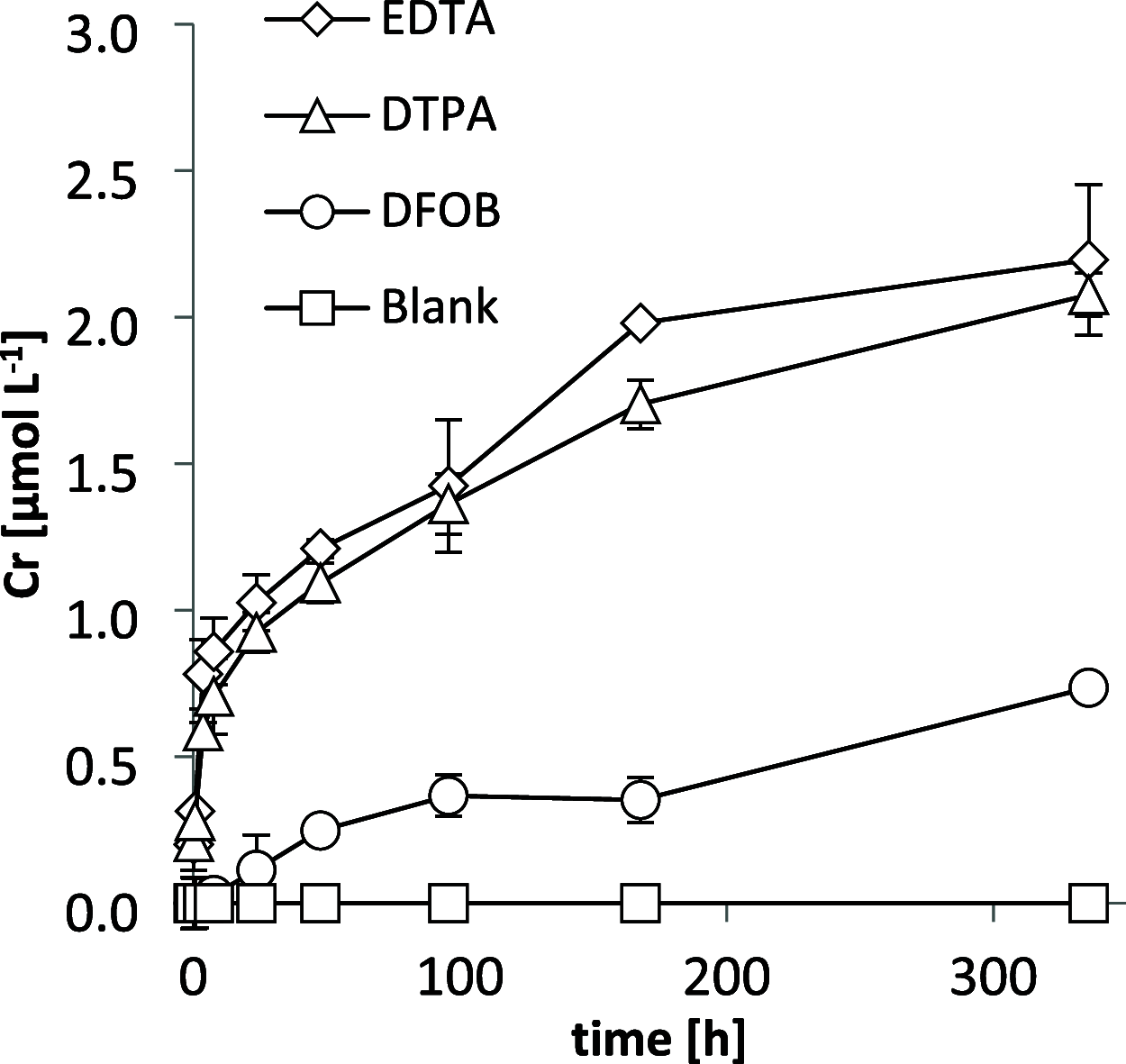
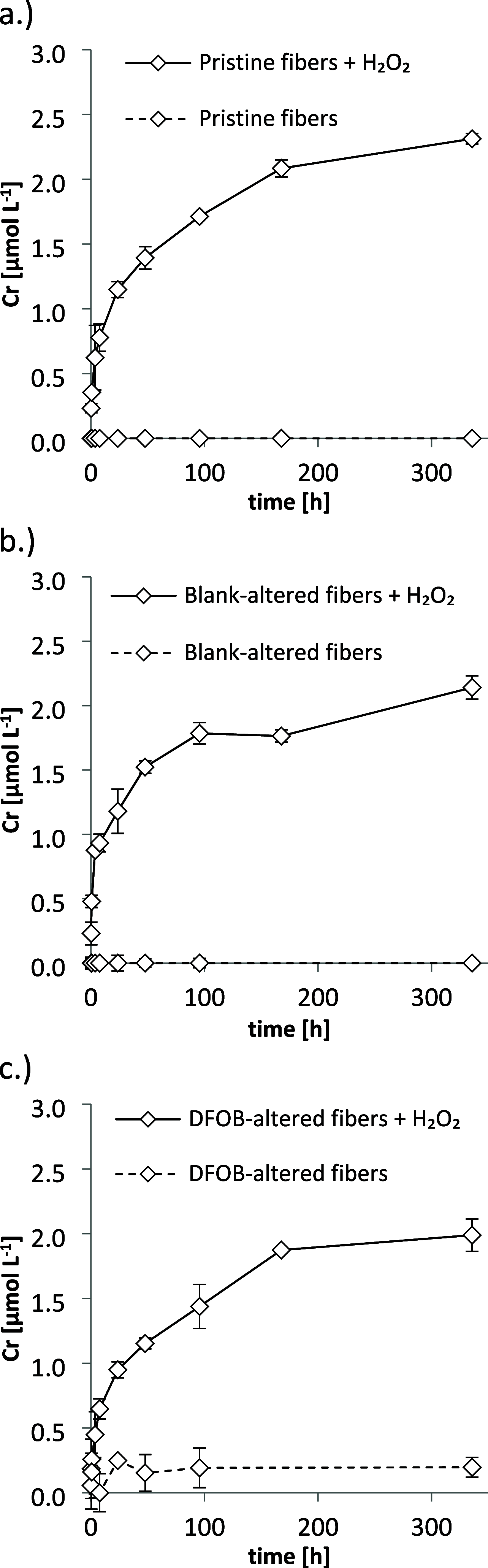
Chrysotile asbestos is a carcinogenic mineral that has abundantly been used in industrial and consumer applications. The carcinogenicity of the fibers is partly governed by reactive Fe surface sites that catalyze the generation of highly toxic hydroxyl radicals (HO•) from extracellular hydrogen peroxide (H2O2). Chrysotile also contains Cr, typically in the low mass permille range. In this study, we examined the leaching of Cr from fibers at the physiological lung pH of 7.4 in the presence and absence of H2O2. Furthermore, we investigated the potential of cells from typical asbestos-burdened tissues and cancers to take up Cr leached from chrysotile in PCR expression, immunoblot, and cellular Cr uptake experiments. Finally, the contribution of Cr to fiber-mediated H2O2 decomposition and HO• generation was studied. Chromium readily dissolved from chrysotile fibers in its genotoxic and carcinogenic hexavalent redox state upon oxidation by H2O2. Lung epithelial, mesothelial, lung carcinoma, and mesothelioma cells expressed membrane-bound Cr(VI) transporters and accumulated Cr up to 10-fold relative to the Cr(VI) concentration in the spiked medium. Conversely, anion transporter inhibitors decreased cellular Cr(VI) uptake up to 45-fold. Finally, chromium associated with chrysotile neither decomposed H2O2 nor contributed to fiber-mediated HO• generation. Altogether, our results support the hypothesis that Cr may leach from inhaled chrysotile in its hexavalent state and subsequently accumulate in cells of typically asbestos-burdened tissues, which could contribute to the carcinogenicity of chrysotile fibers. However, unlike Fe, Cr did not significantly contribute to the adverse radical production of chrysotile.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



References
-
- Catherine H.; Skinner W. Mineralogy of asbestos minerals. Indoor Built Environ. 2003, 12, 385–389. 10.1177/1420326X03037003. - DOI
-
- Oury T. D., Sporn T. A., Roggli V. L., and (Editors). (2014) Pathology of asbestos associated diseases, 10.1007/978-3-642-41193-9. - DOI
-
- Frank A.The History of the Extraction and Uses of Asbestos. 1-7. 2005.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

