Functionally integrating nanoparticles alleviate deep vein thrombosis in pregnancy and rescue intrauterine growth restriction
- PMID: 36418325
- PMCID: PMC9684510
- DOI: 10.1038/s41467-022-34878-2
Functionally integrating nanoparticles alleviate deep vein thrombosis in pregnancy and rescue intrauterine growth restriction
Abstract
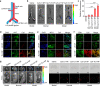
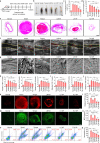
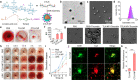
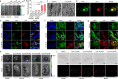
There is still unmet demand for effective, safe, and patient-friendly anti-thrombotics to treat deep vein thrombosis (DVT) during pregnancy. Here we first engineer a bioactive amphiphile (TLH) by simultaneously conjugating Tempol and linoleic acid onto low molecular weight heparin (LMWH), which can assemble into multifunctional nanoparticles (TLH NP). In pregnant rats with DVT, TLH NP can target and dissolve thrombi, recanalize vessel occlusion, and eradicate the recurrence of thromboembolism, thereby reversing DVT-mediated intrauterine growth restriction and delayed development of fetuses. Mechanistically, therapeutic effects of TLH NP are realized by inhibiting platelet aggregation, facilitating thrombolysis, reducing local inflammation, attenuating oxidative stress, promoting endothelial repair, and increasing bioavailability. By decorating with a fibrin-binding peptide, targeting efficiency and therapeutic benefits of TLH NP are considerably improved. Importantly, LMWH nanotherapies show no toxicities to the mother and fetus at the dose 10-time higher than the examined therapeutic dosage.
© 2022. The Author(s).
Conflict of interest statement
H.B.Q., J.C., S.Q.Z., and X.Y.J. are inventors in a pending patent filed by the China National Intellectual Property Administration (No. 202210798192.X, July 06 2022) related to TLH NP and CTLH NP, but the rights belong to the Women and Children’s Hospital of Chongqing Medical University. All other authors declare that they have no competing interests.
Figures





Similar articles
-
Pregnancy after catheter-directed thrombolysis for acute iliofemoral deep venous thrombosis.Phlebology. 2013 Mar;28 Suppl 1:34-8. doi: 10.1177/0268355513477286. Phlebology. 2013. PMID: 23482532
-
Low-molecular-weight heparin (LMWH) in the treatment of thrombosis.Eur J Med Res. 2004 Apr 30;9(4):225-39. Eur J Med Res. 2004. PMID: 15210403 Review.
-
Out of hospital treatment with subcutaneous low molecular weight heparin in patients with acute deep-vein thrombosis: a prospective study in daily practice.Haematologica. 2006 Aug;91(8):1052-8. Haematologica. 2006. PMID: 16885045
-
Home therapy with LMWH in deep vein thrombosis: randomized study comparing single and double daily administrations.Angiology. 2007 Jun-Jul;58(3):316-22. doi: 10.1177/0003319707301757. Angiology. 2007. PMID: 17626986 Clinical Trial.
-
Initial treatment of venous thromboembolism.Circulation. 2004 Aug 31;110(9 Suppl 1):I3-9. doi: 10.1161/01.CIR.0000140904.52752.0c. Circulation. 2004. PMID: 15339875 Review.
Cited by
-
Oxidative stress in vascular surgical diseases: mechanisms, impacts and therapeutic perspectives.Front Pharmacol. 2025 Apr 9;16:1527684. doi: 10.3389/fphar.2025.1527684. eCollection 2025. Front Pharmacol. 2025. PMID: 40271068 Free PMC article. Review.
-
Actively Targeted Nanomedicines: A New Perspective for the Treatment of Pregnancy-Related Diseases.Reprod Sci. 2024 Sep;31(9):2560-2575. doi: 10.1007/s43032-024-01520-z. Epub 2024 Mar 29. Reprod Sci. 2024. PMID: 38553575 Review.
-
Untethered miniature robots for minimally invasive thrombus treatment: From bench to clinical trials.Innovation (Camb). 2025 Mar 11;6(6):100874. doi: 10.1016/j.xinn.2025.100874. eCollection 2025 Jun 2. Innovation (Camb). 2025. PMID: 40528888 Free PMC article. Review.
-
Nanomicrosphere sustained-release urokinase systems with antioxidant properties for deep vein thrombosis therapy.RSC Adv. 2024 Feb 28;14(10):7195-7205. doi: 10.1039/d3ra07221e. eCollection 2024 Feb 21. RSC Adv. 2024. PMID: 38419677 Free PMC article.
-
Exploring metabolite-mediated links between lipidome and deep vein thrombosis: Insights from Mendelian randomization analysis.Medicine (Baltimore). 2025 Mar 7;104(10):e41783. doi: 10.1097/MD.0000000000041783. Medicine (Baltimore). 2025. PMID: 40068057 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

