Redox Regulation of Signaling Complex between Caveolin-1 and Neuronal Calcium Sensor Recoverin
- PMID: 36421712
- PMCID: PMC9687869
- DOI: 10.3390/biom12111698
Redox Regulation of Signaling Complex between Caveolin-1 and Neuronal Calcium Sensor Recoverin
Abstract
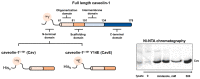
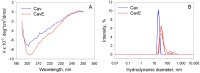
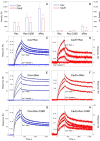
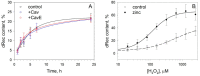
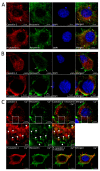
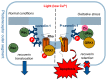
Caveolin-1 is a cholesterol-binding scaffold protein, which is localized in detergent-resistant membrane (DRM) rafts and interacts with components of signal transduction systems, including visual cascade. Among these components are neuronal calcium sensors (NCSs), some of which are redox-sensitive proteins that respond to calcium signals by modulating the activity of multiple intracellular targets. Here, we report that the formation of the caveolin-1 complex with recoverin, a photoreceptor NCS serving as the membrane-binding regulator of rhodopsin kinase (GRK1), is a redox-dependent process. Biochemical and biophysical in vitro experiments revealed a two-fold decreased affinity of recoverin to caveolin-1 mutant Y14E mimicking its oxidative stress-induced phosphorylation of the scaffold protein. At the same time, wild-type caveolin-1 demonstrated a 5-10-fold increased affinity to disulfide dimer of recoverin (dRec) or its thiol oxidation mimicking the C39D mutant. The formation of dRec in vitro was not affected by caveolin-1 but was significantly potentiated by zinc, the well-known mediator of redox homeostasis. In the MDCK cell model, oxidative stress indeed triggered Y14 phosphorylation of caveolin-1 and disulfide dimerization of recoverin. Notably, oxidative conditions promoted the accumulation of phosphorylated caveolin-1 in the plasma membrane and the recruitment of recoverin to the same sites. Co-localization of these proteins was preserved upon depletion of intracellular calcium, i.e., under conditions reducing membrane affinity of recoverin but favoring its interaction with caveolin-1. Taken together, these data suggest redox regulation of the signaling complex between recoverin and caveolin-1. During oxidative stress, the high-affinity interaction of thiol-oxidized recoverin with caveolin-1/DRMs may disturb the light-induced translocation of the former within photoreceptors and affect rhodopsin desensitization.
Keywords: apoptosis; caveolin-1; oxidative stress; photoreceptor; recoverin; retina.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Photoreceptor calcium sensor proteins in detergent-resistant membrane rafts are regulated via binding to caveolin-1.Cell Calcium. 2018 Jul;73:55-69. doi: 10.1016/j.ceca.2018.04.003. Epub 2018 Apr 14. Cell Calcium. 2018. PMID: 29684785
-
Light-Induced Thiol Oxidation of Recoverin Affects Rhodopsin Desensitization.Front Mol Neurosci. 2019 Jan 7;11:474. doi: 10.3389/fnmol.2018.00474. eCollection 2018. Front Mol Neurosci. 2019. PMID: 30666186 Free PMC article.
-
Light-induced disulfide dimerization of recoverin under ex vivo and in vivo conditions.Free Radic Biol Med. 2015 Jun;83:283-95. doi: 10.1016/j.freeradbiomed.2015.03.001. Epub 2015 Mar 12. Free Radic Biol Med. 2015. PMID: 25772009
-
Dimerization of Neuronal Calcium Sensor Proteins.Front Mol Neurosci. 2018 Nov 2;11:397. doi: 10.3389/fnmol.2018.00397. eCollection 2018. Front Mol Neurosci. 2018. PMID: 30450035 Free PMC article. Review.
-
Ca2+-dependent control of rhodopsin phosphorylation: recoverin and rhodopsin kinase.Adv Exp Med Biol. 2002;514:69-99. doi: 10.1007/978-1-4615-0121-3_5. Adv Exp Med Biol. 2002. PMID: 12596916 Review.
Cited by
-
Redox Regulation of Protein Functioning.Biomolecules. 2023 Aug 22;13(9):1281. doi: 10.3390/biom13091281. Biomolecules. 2023. PMID: 37759681 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

