Liraglutide Effectiveness in Type 2 Diabetes: Insights from a Real-World Cohort of Portuguese Patients
- PMID: 36422260
- PMCID: PMC9694138
- DOI: 10.3390/metabo12111121
Liraglutide Effectiveness in Type 2 Diabetes: Insights from a Real-World Cohort of Portuguese Patients
Abstract
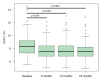
Liraglutide is a long-acting glucagon-like peptide-1 receptor agonist prescribed to diabetic patients for glycaemic control. To understand the impact of liraglutide in the real-world setting, this study analysed its effects in a Portuguese cohort of Type 2 diabetes patients. This was an observational, multicentric, and retrospective study that included 191 liraglutide-treated patients with at least 12 months of treatment. Patients’ data were collected and analysed during a 24-month follow-up period. Overall, liraglutide treatment effectively reduced HbA1c levels from 8.3% to around 7.5%, after 6, 12, and 24 months (p < 0.001). In fact, 38.2%, 37.2%, and 44.8% of patients at 6, 12, and 24 months, respectively, experienced an HbA1c reduction of at least 1%. Moreover, a persistent reduction in anthropometric features was also observed, with 44.0%, 47.6%, and 54.4% of patients achieving a weight reduction of at least 3% at 6, 12, and 24 months, respectively. Finally, significant improvements were observed in the HDL-c and LDL-c levels. Our results demonstrate that liraglutide effectively promoted the reduction of HbA1c values during routine clinical practice, which was sustained throughout the study. In addition, there were significant improvements in anthropometric parameters and other cardiovascular risk factors.
Keywords: anthropometric parameters; cardiovascular risk factors; glycaemic control; liraglutide; real-world evidence; type 2 diabetes.
Conflict of interest statement
J.S.-N. has received research support or honoraria for consultancy or training activities from Abbott, AstraZeneca, Bial, Boehringer Ingelheim, Lilly, Janssen Pharmaceuticals, Medinfar, Merck SA, Merck Sharp & Dohme, Mundipharma, Novartis Pharmaceuticals, Novo Nordisk, Roche, Sanofi, Servier, and Tecnimede. E.N. has received research grants and/or honoraria as a consultant, member of advisory boards, or speaker from AstraZeneca, Bial, LifeScan, Merck Sharp & Dohme, and Novo Nordisk. J.L. has received research grants and/or honoraria as a consultant, member of advisory boards, or speaker from AstraZeneca, Boehringer Ingelheim, Lilly, Medinfar, Merck Sharp & Dohme, and Novo Nordisk. J.D. has received speaker honoraria from or has participated in advisory boards for Abbott, Amgen, AstraZeneca, Boehringer Ingelheim, Lilly, Merck Sharp & Dohme, Mundipharma, Novo Nordisk, and Sanofi. A.G.-F. has received honoraria as a consultant/speaker from Medinfar. M.A. has received honoraria as a consultant/speaker from AstraZeneca and Medinfar. S.B.S. has received research grants and/or honoraria as a consultant, member of advisory boards, or speaker from Bial, Boehringer Ingelheim, Lilly, Medinfar, Merck Sharp & Dohme, and Novo Nordisk. N.C. has received honoraria as a consultant/speaker from Lilly and Novo Nordisk. R.D. has received research grants and/or honoraria as a consultant, member of advisory boards, or speaker from Abbott, AstraZeneca, Boehringer Ingelheim, Lilly, Medinfar, Merck Sharp & Dohme, Novartis, Novo Nordisk, Tecnimede, and Sanofi. J.F.R. has received honoraria for consultancy from Abbott, AstraZeneca, Boehringer Ingelheim, Lilly, Merck Sharp & Dohme, Novo Nordisk, and Roche.
Figures
Similar articles
-
Long-term effectiveness and safety of liraglutide in clinical practice.Minerva Endocrinol. 2013 Mar;38(1):103-12. Minerva Endocrinol. 2013. PMID: 23435446
-
Long-Term Effectiveness of Liraglutide in Association with Patients' Baseline Characteristics in Real-Life Setting in Croatia: An Observational, Retrospective, Multicenter Study.Diabetes Ther. 2017 Dec;8(6):1297-1308. doi: 10.1007/s13300-017-0324-x. Epub 2017 Oct 26. Diabetes Ther. 2017. PMID: 29076038 Free PMC article.
-
Long-Term clinical efficacy of liraglutide for type 2 diabetes: real-world evidence and outcomes from Pakistan.J Pharm Policy Pract. 2024 Dec 4;17(1):2432462. doi: 10.1080/20523211.2024.2432462. eCollection 2024. J Pharm Policy Pract. 2024. PMID: 39640418 Free PMC article.
-
Clinical Effectiveness of Liraglutide in Type 2 Diabetes Treatment in the Real-World Setting: A Systematic Literature Review.Diabetes Ther. 2016 Sep;7(3):411-38. doi: 10.1007/s13300-016-0180-0. Epub 2016 Jun 27. Diabetes Ther. 2016. PMID: 27350545 Free PMC article. Review.
-
Glycaemic efficacy of glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors as add-on therapy to metformin in subjects with type 2 diabetes-a review and meta analysis.Diabetes Obes Metab. 2012 Aug;14(8):762-7. doi: 10.1111/j.1463-1326.2012.01603.x. Epub 2012 Apr 24. Diabetes Obes Metab. 2012. PMID: 22471248 Review.
References
-
- Stratton I.M., Adler A.I., Neil H.A., Matthews D.R., Manley S.E., Cull C.A., Hadden D., Turner R.C., Holman R.R. Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): Prospective observational study. BMJ. 2000;321:405–412. doi: 10.1136/bmj.321.7258.405. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

