Synthesis and Characterisation of Novel Bis(diphenylphosphane oxide)methanidoytterbium(III) Complexes
- PMID: 36431801
- PMCID: PMC9698226
- DOI: 10.3390/molecules27227704
Synthesis and Characterisation of Novel Bis(diphenylphosphane oxide)methanidoytterbium(III) Complexes
Abstract
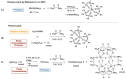
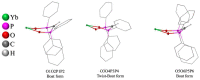
Reaction of [YbCp2(dme)] (Cp = cyclopentadienyl, dme = 1,2 dimethoxyethane) with bis(diphenylphosphano)methane dioxide (H2dppmO2) leads to deprotonation of the ligand H2dppmO2 and oxidation of ytterbium, forming an extremely air-sensitive product, [YbIII(HdppmO2)3] (1), a six-coordinate complex with three chelating (OPCHPO) HdppmO2 ligands. Complex 1 was also obtained by a redox transmetallation/protolysis synthesis from metallic ytterbium, Hg(C6F5)2, and H2dppmO2. In a further preparation, the reaction of [Yb(C6F5)2] with H2dppmO2, not only yielded compound 1, but also gave a remarkable tetranuclear cage, [Yb4(µ-HdppmO2)6(µ-F)6] (2) containing two [Yb(µ-F)]2 rhombic units linked by two fluoride ligands and the tetranuclear unit is encapsulated by six bridging HdppmO2 donors. The fluoride ligands of the cage result from C-F activation of pentafluorobenzene and concomitant formation of p-H2C6F4 and m-H2C6F4, the last being an unexpected product.
Keywords: C-F activation; fluoride cage; methanide; phosphine oxide; ytterbium.
Conflict of interest statement
The authors declare no conflict of interest.
Figures








References
-
- Hart F.A. Comprehensive Coordination Chemistry. Volume 3 Pergamon Press; Oxford, UK: 1987. Scandium, Yttrium and the lanthanides, Chapter 39.
-
- Cotton S. Comprehensive Coordination Chemistry II. Volume 3 Elsevier; Amsterdam, The Netherlands: 2003. Scandium, Yttrium and the Lanthanides, Chapter 3.2, Section 3.2.2.5.2.
-
- Guino-o M.A., Bustrom B., Tigaa R.A., de Bettencourt-Dias A. Microwave-assisted synthesis of ternary lanthanide(2-thenoyltrifluoroacetone)3(triphenylphosphine oxide)2 complexes. Inorg. Chim. Acta. 2017;464:23–30. doi: 10.1016/j.ica.2017.04.021. - DOI
-
- Platt A.W.G. Lanthanide phosphine oxide complexes. Coord. Chem. Rev. 2017;340:62–78. doi: 10.1016/j.ccr.2016.09.012. - DOI
-
- Moeller T. Gmelin Handbook of Inorganic Chemistry. Springer; Berlin/Heidelberg, Germany: 1983. System 39, Part D6.
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

