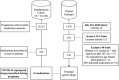
A medication-wide association study (MWAS) on repurposed drugs for COVID-19 with Pre-pandemic prescription medication exposure and pregnancy outcomes
- PMID: 36433981
- PMCID: PMC9700703
- DOI: 10.1038/s41598-022-24218-1
A medication-wide association study (MWAS) on repurposed drugs for COVID-19 with Pre-pandemic prescription medication exposure and pregnancy outcomes
Abstract
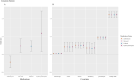
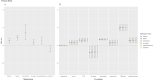
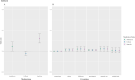
Information on effects of medication therapies during pregnancy is lacking as pregnant patients are often excluded from clinical trials. This retrospective study explores the potential of using electronic health record (EHR) data to inform safety profiles of repurposed COVID medication therapies on pregnancy outcomes using pre-COVID data. We conducted a medication-wide association study (MWAS) on prescription medication exposures during pregnancy and the risk of cesarean section, preterm birth, and stillbirth, using EHR data between 2010-2017 on deliveries at PennMedicine. Repurposed drugs studied for treatment of COVID-19 were extracted from ClinicalTrials.gov (n = 138). We adjusted for known comorbidities diagnosed within 2 years prior to birth. Using previously developed medication mapping and delivery-identification algorithms, we identified medication exposure in 2,830 of a total 63,334 deliveries; from 138 trials, we found 31 medications prescribed and included in our cohort. We found 21 (68%) of the 31 medications were not positively associated with increased risk of the outcomes examined. With caution, these medications warrant potential for inclusion of pregnant individuals in future studies, while drugs found to be associated with pregnancy outcomes require further investigation. MWAS facilitates hypothesis-driven evaluation of drug safety across all prescription medications, revealing potential drug candidates for further research.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Prenatal Health Care Outcomes Before and During the COVID-19 Pandemic Among Pregnant Individuals and Their Newborns in an Integrated US Health System.JAMA Netw Open. 2023 Jul 3;6(7):e2324011. doi: 10.1001/jamanetworkopen.2023.24011. JAMA Netw Open. 2023. PMID: 37462973 Free PMC article.
-
The impact of covid-19 pandemic on pregnancy outcome.BMC Pregnancy Childbirth. 2023 Nov 22;23(1):811. doi: 10.1186/s12884-023-06098-z. BMC Pregnancy Childbirth. 2023. PMID: 37993814 Free PMC article.
-
Obstetrical outcomes and maternal morbidities associated with COVID-19 in pregnant women in France: A national retrospective cohort study.PLoS Med. 2021 Nov 30;18(11):e1003857. doi: 10.1371/journal.pmed.1003857. eCollection 2021 Nov. PLoS Med. 2021. PMID: 34847147 Free PMC article.
-
Efficacy and safety of therapies for COVID-19 in pregnancy: a systematic review and meta-analysis.BMC Infect Dis. 2023 Nov 9;23(1):776. doi: 10.1186/s12879-023-08747-2. BMC Infect Dis. 2023. PMID: 37946100 Free PMC article.
-
Outcome of coronavirus spectrum infections (SARS, MERS, COVID-19) during pregnancy: a systematic review and meta-analysis.Am J Obstet Gynecol MFM. 2020 May;2(2):100107. doi: 10.1016/j.ajogmf.2020.100107. Epub 2020 Mar 25. Am J Obstet Gynecol MFM. 2020. PMID: 32292902 Free PMC article.
References
-
- Figueiro-Filho, E. A., Yudin, M., Farine, D. COVID-19 during pregnancy: an overview of maternal characteristics, clinical symptoms, maternal and neonatal outcomes of 10,996 cases described in 15 countries. J. Perinat. Med. (2020). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

