Sustained delivery of CpG oligodeoxynucleotide by acetalated dextran microparticles augments effector response to Computationally Optimized Broadly Reactive Antigen (COBRA) influenza hemagglutinin
- PMID: 36436743
- PMCID: PMC9789738
- DOI: 10.1016/j.ijpharm.2022.122429
Sustained delivery of CpG oligodeoxynucleotide by acetalated dextran microparticles augments effector response to Computationally Optimized Broadly Reactive Antigen (COBRA) influenza hemagglutinin
Abstract
A subunit or protein-based influenza vaccine can be a safer alternative to live attenuated vaccine (Flumist) and require fewer boosts than an inactivated vaccine (e.g. Fluzone). However, to form an effective subunit vaccine, an adjuvant is often needed. In this work we used electrospray to encapsulate the hydrophilic adjuvant CpG into microparticles made from the hydrophobic biodegradable polymer acetalated dextran. To understand the rate of particle degradation on CpG release, polymer that was slow (21 h at phagosomal pH 5) and fast (0.25 h at pH 5) degrading was used to encapsulate the adjuvant. The slow-degrading particles exhibited the greatest degree of innate immune stimulation of antigen-presenting cells in vitro. In mice, the broadly acting Computationally Optimized Broadly Reactive Antigen (COBRA) Y2 influenza hemagglutinin (HA) antigen was used with CpG particles, soluble CpG, or MF-59 like adjuvant Addavax. Particles and soluble CpG elicited similar induction of anti-HA antibodies and protection against lethal influenza challenge, but the sustained release particles elicited the highest levels antibody effector functions. These results demonstrate a suitable method for encapsulation of CpG oligonucleotide in a hydrophobic particle matrix, and suggest that sustained release of CpG from Ace-DEX microparticles could potentially be used to induce potent antibody effector functions.
Keywords: Intramuscular injection; Nanoparticle vaccine; Seasonal influenza vaccine; Tunable degradation; Universal influenza vaccine.
Copyright © 2022 Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures

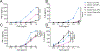
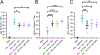

Similar articles
-
Ethoxy Acetalated Dextran-Based Biomaterials for Therapeutic Applications.Polymers (Basel). 2024 Sep 29;16(19):2756. doi: 10.3390/polym16192756. Polymers (Basel). 2024. PMID: 39408467 Free PMC article. Review.
-
Multi-COBRA hemagglutinin formulated with cGAMP microparticles elicits protective immune responses against influenza viruses.mSphere. 2024 Jul 30;9(7):e0016024. doi: 10.1128/msphere.00160-24. Epub 2024 Jun 26. mSphere. 2024. PMID: 38920382 Free PMC article.
-
Optimizing delivery in a multivalent subunit influenza vaccine using mixed polymeric microparticle degradation rates.J Control Release. 2025 Aug 10;384:113936. doi: 10.1016/j.jconrel.2025.113936. Epub 2025 Jun 6. J Control Release. 2025. PMID: 40482923
-
Vinyl Sulfone-functionalized Acetalated Dextran Microparticles as a Subunit Broadly Acting Influenza Vaccine.AAPS J. 2023 Jan 31;25(1):22. doi: 10.1208/s12248-023-00786-6. AAPS J. 2023. PMID: 36720729 Free PMC article.
-
Next-Generation Influenza HA Immunogens and Adjuvants in Pursuit of a Broadly Protective Vaccine.Viruses. 2021 Mar 24;13(4):546. doi: 10.3390/v13040546. Viruses. 2021. PMID: 33805245 Free PMC article. Review.
Cited by
-
Ethoxy Acetalated Dextran-Based Biomaterials for Therapeutic Applications.Polymers (Basel). 2024 Sep 29;16(19):2756. doi: 10.3390/polym16192756. Polymers (Basel). 2024. PMID: 39408467 Free PMC article. Review.
-
Natural Polymeric Composites Derived from Animals, Plants, and Microbes for Vaccine Delivery and Adjuvant Applications: A Review.Gels. 2023 Mar 15;9(3):227. doi: 10.3390/gels9030227. Gels. 2023. PMID: 36975676 Free PMC article. Review.
-
Delivery of small molecule mast cell activators for West Nile Virus vaccination using acetalated dextran microparticles.Int J Pharm. 2023 Mar 5;634:122658. doi: 10.1016/j.ijpharm.2023.122658. Epub 2023 Jan 30. Int J Pharm. 2023. PMID: 36731641 Free PMC article.
References
-
- Belongia EA, Simpson MD, King JP, Sundaram ME, Kelley NS, Osterholm MT, McLean HQ, 2016. Variable influenza vaccine effectiveness by subtype: a systematic review and meta-analysis of test-negative design studies. Lancet Infect Dis 16, 942–951. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

