Phenomapping in heart failure with preserved ejection fraction: insights, limitations, and future directions
- PMID: 36448685
- PMCID: PMC10144733
- DOI: 10.1093/cvr/cvac179
Phenomapping in heart failure with preserved ejection fraction: insights, limitations, and future directions
Erratum in
-
Erratum to: Phenomapping in heart failure with preserved ejection fraction: insights, limitations, and future directions.Cardiovasc Res. 2023 May 2;119(4):1096. doi: 10.1093/cvr/cvad002. Cardiovasc Res. 2023. PMID: 36680536 Free PMC article. No abstract available.
Abstract
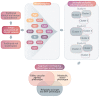
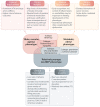
Heart failure with preserved ejection fraction (HFpEF) is a heterogeneous entity with complex pathophysiology and manifestations. Phenomapping is the process of applying statistical learning techniques to patient data to identify distinct subgroups based on patterns in the data. Phenomapping has emerged as a technique with potential to improve the understanding of different HFpEF phenotypes. Phenomapping efforts have been increasing in HFpEF over the past several years using a variety of data sources, clinical variables, and statistical techniques. This review summarizes methodologies and key takeaways from these studies, including consistent discriminating factors and conserved HFpEF phenotypes. We argue that phenomapping results to date have had limited implications for clinical care and clinical trials, given that the phenotypes, as currently described, are not reliably identified in each study population and may have significant overlap. We review the inherent limitations of aggregating and utilizing phenomapping results. Lastly, we discuss potential future directions, including using phenomapping to optimize the likelihood of clinical trial success or to drive discovery in mechanisms of the disease process of HFpEF.
Keywords: Heart failure with preserved ejection fraction; phenomapping; phenotype; phenotyping.
© The Author(s) 2022. Published by Oxford University Press on behalf of the European Society of Cardiology. All rights reserved. For permissions, please email: journals.permissions@oup.com.
Conflict of interest statement
Conflict of interest: Dr Peters is supported by the National Heart Lung and Blood Institute (T32HL069749). Dr Tromp is supported by the National University of Singapore Start-up grant, the tier 1 grant from the ministry of education and the CS-IRG New Investigator Grant from the National Medical Research Council; has received consulting or speaker fees from Daiichi-Sankyo, Boehringer Ingelheim, Roche diagnostics and Us2.ai, owns patent US-10702247-B2 unrelated to the present work. Dr Shah has received research grants from Actelion, AstraZeneca, Corvia, Novartis, and Pfizer; and consulting fees from Abbott, Actelion, AstraZeneca, Amgen, Aria CV, Axon Therapies, Bayer, Boehringer-Ingelheim, Boston Scientific, Bristol-Myers Squibb, Cardiora, Coridea, CVRx, Cyclerion, Cytokinetics, Edwards Lifesciences, Eidos, Eisai, Imara, Impulse Dynamics, Intellia, Ionis, Ironwood, Lilly, Merck, MyoKardia, Novartis, Novo Nordisk, Pfizer, Prothena, Regeneron, Rivus, Sanofi, Shifamed, Tenax, Tenaya, and United Therapeutics. Dr Lam is supported by a Clinician Scientist Award from the National Medical Research Council of Singapore; has received research support from AstraZeneca, Bayer, Boston Scientific and Roche Diangostics; has served as consultant or on the Advisory Board/Steering Committee/Executive Committee for Actelion, Amgen, Applied Therapeutics, AstraZenea, Bayer, Boehringer Ingelheim, Boston Scientific, Cytokinetics, Darma Inc., Janssen Research & Development LLC, Medscape/WebMD Global LLC, Merck, Novartis, Novo Nordisk, Radcliffe Group Ltd., Roche Diagnostics, Sanofi and Us2.ai. Dr Borlaug has received research grants from AstraZeneca, Corvia, Medtronic, GlaxoSmithKline, Mesoblast, Novartis, Tenax Therapeutics, and consulting fees from Actelion, Amgen, Aria, Axon Therapies, Boehringer Ingelheim, Edwards Lifesciences, Eli Lilly, Imbria, Janssen, Merck, Novo Nordisk, and VADovations. Dr Sharma is a consultant and advisory board member of AstraZeneca, Bayer, Boehringer-Ingelheim, Janssen, Novartis, NovoNordisk, and Rivus and receives honoraria; has received research support from Amgen. Dr Pandey received grant funding outside the present study from Applied Therapeutics, Gilead and Myovista; has received honoraria as an advisor/consultant for Tricog Health Inc and Lilly, USA, Roche and has received nonfinancial support from Pfizer and Merck. Dr Kitzman has received honoraria outside the present study as a consultant for Bayer, Merck, Medtronic, Relypsa, Merck, Corvia Medical, Boehringer-Ingelheim, NovoNordisk, AstraZeneca, Rivus, Pfizer, and Novartis; grant funding outside the present study from Novartis, Bayer, NovoNordisk, and AstraZeneca; and has stock ownership in Gilead Sciences. Dr Mentz has received research support and honoraria from Abbott, American Regent, Amgen, AstraZeneca, Bayer, Boehringer Ingelheim/Eli Lilly, Boston Scientific, Cytokinetics, Fast BioMedical, Gilead, Innolife, Medtronic, Merck, Novartis, Relypsa, Respicardia, Roche, Sanofi, Vifor, Windtree Therapeutics, and Zoll. All other authors have reported that they have no relationships relevant to the contents of this paper to disclose.
Figures


References
-
- Pandey A, Shah SJ, Butler J, Kellogg DL, Lewis GD, Forman DE, Mentz RJ, Borlaug BA, Simon MA, Chirinos JA, Fielding RA, Volpi E, Molina AJA, Haykowsky MJ, Sam F, Goodpaster BH, Bertoni AG, Justice JN, White JP, Ding J, Hummel SL, LeBrasseur NK, Taffet GE, Pipinos II, Kitzman D. Exercise intolerance in older adults with heart failure with preserved ejection fraction: JACC state-of-the-art review. J Am Coll Cardiol 2021;78:1166–1187. - PMC - PubMed
-
- Lewis GA, Schelbert EB, Williams SG, Cunnington C, Ahmed F, McDonagh TA, Miller CA. Biological phenotypes of heart failure with preserved ejection fraction. J Am Coll Cardiol 2017;70:2186–2200. - PubMed
-
- Kitzman DW, Shah SJ. The HFpEF obesity phenotype: the elephant in the room. J Am Coll Cardiol 2016;68:200–203. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

