A novel post-developmental role of the Hox genes underlies normal adult behavior
- PMID: 36454751
- PMCID: PMC9894213
- DOI: 10.1073/pnas.2209531119
A novel post-developmental role of the Hox genes underlies normal adult behavior
Abstract
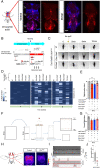
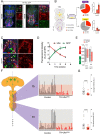
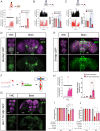
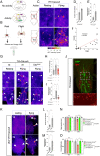
The molecular mechanisms underlying the stability of mature neurons and neural circuits are poorly understood. Here we explore this problem and discover that the Hox genes are a component of the genetic program that maintains normal neural function in adult Drosophila. We show that post-developmental downregulation of the Hox gene Ultrabithorax (Ubx) in adult neurons leads to substantial anomalies in flight. Mapping the cellular basis of these effects reveals that Ubx is required within a subset of dopaminergic neurons, and cell circuitry analyses in combination with optogenetics allow us to link these dopaminergic neurons to flight control. Functional imaging experiments show that Ubx is necessary for normal dopaminergic activity, and neuron-specific RNA-sequencing defines two previously uncharacterized ion channel-encoding genes as potential mediators of Ubx behavioral roles. Our study thus reveals a novel role of the Hox system in controlling adult behavior and neural function. Based on the broad evolutionary conservation of the Hox system across distantly related animal phyla, we predict that the Hox genes might play neurophysiological roles in adult forms of other species, including humans.
Keywords: Drosophila; Hox genes; flight; neuron; post-mitotic.
Conflict of interest statement
The authors declare no competing interest.
Figures

Similar articles
-
A Single MicroRNA-Hox Gene Module Controls Equivalent Movements in Biomechanically Distinct Forms of Drosophila.Curr Biol. 2019 Aug 19;29(16):2665-2675.e4. doi: 10.1016/j.cub.2019.06.082. Epub 2019 Jul 18. Curr Biol. 2019. PMID: 31327720 Free PMC article.
-
The RNA-binding protein ELAV regulates Hox RNA processing, expression and function within the Drosophila nervous system.Development. 2014 May;141(10):2046-56. doi: 10.1242/dev.101519. Development. 2014. PMID: 24803653 Free PMC article.
-
MicroRNA-dependent regulation of Hox gene expression sculpts fine-grain morphological patterns in a Drosophila appendage.Development. 2018 Oct 16;145(20):dev161133. doi: 10.1242/dev.161133. Development. 2018. PMID: 30143542 Free PMC article.
-
The bithorax complex of Drosophila an exceptional Hox cluster.Curr Top Dev Biol. 2009;88:1-33. doi: 10.1016/S0070-2153(09)88001-0. Curr Top Dev Biol. 2009. PMID: 19651300 Review.
-
Hox miRNA regulation within the Drosophila Bithorax complex: Patterning behavior.Mech Dev. 2015 Nov;138 Pt 2(0 2):151-159. doi: 10.1016/j.mod.2015.08.006. Epub 2015 Aug 23. Mech Dev. 2015. PMID: 26311219 Free PMC article. Review.
Cited by
-
Epigenome-wide DNA methylation analysis of late-stage mild cognitive impairment.Front Cell Dev Biol. 2024 Jan 16;12:1276288. doi: 10.3389/fcell.2024.1276288. eCollection 2024. Front Cell Dev Biol. 2024. PMID: 38298218 Free PMC article.
-
Maintenance of neuronal identity in C. elegans and beyond: Lessons from transcription and chromatin factors.Semin Cell Dev Biol. 2024 Feb 15;154(Pt A):35-47. doi: 10.1016/j.semcdb.2023.07.001. Epub 2023 Jul 11. Semin Cell Dev Biol. 2024. PMID: 37438210 Free PMC article. Review.
-
On growth and form of animal behavior.Front Integr Neurosci. 2025 Feb 4;18:1476233. doi: 10.3389/fnint.2024.1476233. eCollection 2024. Front Integr Neurosci. 2025. PMID: 39967809 Free PMC article.
-
Hox genes in development and beyond.Development. 2023 Jan 1;150(1):dev192476. doi: 10.1242/dev.192476. Epub 2023 Jan 16. Development. 2023. PMID: 36645372 Free PMC article.
-
Hox gene-specific cellular targeting using split intein Trojan exons.Proc Natl Acad Sci U S A. 2024 Apr 23;121(17):e2317083121. doi: 10.1073/pnas.2317083121. Epub 2024 Apr 11. Proc Natl Acad Sci U S A. 2024. PMID: 38602904 Free PMC article.
References
-
- Holmberg J., Perlmann T., Maintaining differentiated cellular identity. Nat. Rev. Genet. 13, 429–439 (2012). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

