Transcriptional control of cone photoreceptor diversity by a thyroid hormone receptor
- PMID: 36454759
- PMCID: PMC9894165
- DOI: 10.1073/pnas.2209884119
Transcriptional control of cone photoreceptor diversity by a thyroid hormone receptor
Abstract
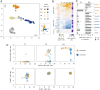
Cone photoreceptor diversity allows detection of wavelength information in light, the first step in color (chromatic) vision. In most mammals, cones express opsin photopigments for sensitivity to medium/long (M, "green") or short (S, "blue") wavelengths and are differentially arrayed over the retina. Cones appear early in retinal neurogenesis but little is understood of the subsequent control of diversity of these postmitotic neurons, because cone populations are sparse and, apart from opsins, poorly defined. It is also a challenge to distinguish potentially subtle differences between cell subtypes within a lineage. Therefore, we derived a Cre driver to isolate individual M and S opsin-enriched cones, which are distributed in counter-gradients over the mouse retina. Fine resolution transcriptome analyses identified expression gradients for groups of genes. The postnatal emergence of gradients indicated divergent differentiation of cone precursors during maturation. Using genetic tagging, we demonstrated a role for thyroid hormone receptor β2 (TRβ2) in control of gradient genes, many of which are enriched for TRβ2 binding sites and TRβ2-regulated open chromatin. Deletion of TRβ2 resulted in poorly distinguished cones regardless of retinal location. We suggest that TRβ2 controls a bipotential transcriptional state to promote cone diversity and the chromatic potential of the species.
Keywords: THRB; color vision; cone photoreceptor; retina; thyroid hormone receptor.
Conflict of interest statement
The authors declare no competing interest.
Figures




References
-
- Nathans J., The evolution and physiology of human color vision: Insights from molecular genetic studies of visual pigments. Neuron 24, 299–312 (1999). - PubMed
-
- Hunt D. M., Peichl L., S cones: Evolution, retinal distribution, development, and spectral sensitivity. Vis. Neurosci. 31, 115–138 (2014). - PubMed
-
- Roorda A., Williams D. R., The arrangement of the three cone classes in the living human eye. Nature 397, 520–522 (1999). - PubMed
-
- Xiao M., Hendrickson A., Spatial and temporal expression of short, long/medium, or both opsins in human fetal cones. J. Comp. Neurol. 425, 545–559 (2000). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

