Association of Placental Growth Factor and Angiopoietin in Human Retinal Endothelial Cell-Pericyte co-Cultures and iPSC-Derived Vascular Organoids
- PMID: 36458540
- PMCID: PMC10417885
- DOI: 10.1080/02713683.2022.2149808
Association of Placental Growth Factor and Angiopoietin in Human Retinal Endothelial Cell-Pericyte co-Cultures and iPSC-Derived Vascular Organoids
Abstract
Purpose: Placental growth factor (PlGF) and Angiopoietin (Ang)-1 are two proteins that are involved in the regulation of endothelial cell (EC) growth and vasculature formation. In the retina and endothelial cells, pericytes are the major source of both molecules. The purpose of this study is to examine the association of PlGF and Ang-1 with human EC/pericyte co-cultures and iPSC-derived vascular organoids.
Methods: In this study, we used co-cultures of human primary retinal endothelial cells (HREC) and primary human retinal pericytes (HRP), western blotting, immunofluorescent analysis, TUNEL staining, LDH-assays, and RNA seq analysis, as well as human-induced pluripotent stem cells (iPSC), derived organoids (VO) to study the association between PlGF and Ang-1.
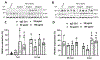
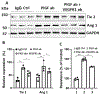
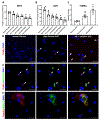
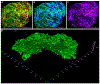
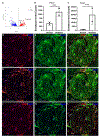
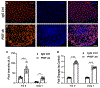
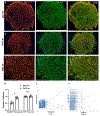
Results: Inhibition of PlGF by PlGF neutralizing antibody in HREC-HRP co-cultures resulted in the increased expression of Ang-1 and Tie-2 in a dose-dependent manner. This upregulation was not observed in HREC and HRP monocultures but only in co-cultures suggesting the association of pericytes and endothelial cells. Furthermore, Vascular endothelial growth factor receptor 1 (VEGFR1) inhibition abolished the Ang-1 and Tie-2 upregulation by PlGF inhibition. The pericyte viability in high-glucose conditions was also reduced by VEGFR1 neutralization. Immunofluorescent analysis showed that Ang-1 and Ang-2 were expressed mainly by perivascular cells in the VO. RNA seq analysis of the RNA isolated from VO in high glucose conditions indicated increased PlGF and Ang-2 expressions in the VO. PlGF inhibition increased the expression of Ang-1 and Tie-2 in VO, increasing the pericyte coverage of the VO microvascular network.
Conclusion: Combined, these results suggest PlGF's role in the regulation of Ang-1 and Tie-2 expression through VEGFR1. These findings provide new insights into the neovascularization process in diabetic retinopathy and new targets for potential therapeutic intervention.
Keywords: PlGF; angiopoietin; diabetic retinopathy; pericyte; vascular organoids.
Conflict of interest statement
None of the authors has a conflict of interest with or source of funding for the submission.
Figures

Similar articles
-
A human retinal microvascular endothelial-pericyte co-culture model to study diabetic retinopathy in vitro.Exp Eye Res. 2020 Dec;201:108293. doi: 10.1016/j.exer.2020.108293. Epub 2020 Oct 8. Exp Eye Res. 2020. PMID: 33039459
-
Connection of pericyte-angiopoietin-Tie-2 system in diabetic retinopathy: friend or foe?Future Med Chem. 2012 Nov;4(17):2163-76. doi: 10.4155/fmc.12.170. Future Med Chem. 2012. PMID: 23190105
-
The angiopoietin/Tie-2 system regulates pericyte survival and recruitment in diabetic retinopathy.Invest Ophthalmol Vis Sci. 2008 May;49(5):2163-71. doi: 10.1167/iovs.07-1206. Invest Ophthalmol Vis Sci. 2008. PMID: 18436850
-
Pericyte-Endothelial Interactions in the Retinal Microvasculature.Int J Mol Sci. 2020 Oct 8;21(19):7413. doi: 10.3390/ijms21197413. Int J Mol Sci. 2020. PMID: 33049983 Free PMC article. Review.
-
VEGFR1 signaling in retinal angiogenesis and microinflammation.Prog Retin Eye Res. 2021 Sep;84:100954. doi: 10.1016/j.preteyeres.2021.100954. Epub 2021 Feb 25. Prog Retin Eye Res. 2021. PMID: 33640465 Free PMC article. Review.
Cited by
-
Application of patient-derived induced pluripotent stem cells and organoids in inherited retinal diseases.Stem Cell Res Ther. 2023 Nov 27;14(1):340. doi: 10.1186/s13287-023-03564-5. Stem Cell Res Ther. 2023. PMID: 38012786 Free PMC article. Review.
-
IC100, a humanized therapeutic monoclonal anti-ASC antibody alleviates oxygen-induced retinopathy in mice.Angiogenesis. 2024 Aug;27(3):423-440. doi: 10.1007/s10456-024-09917-9. Epub 2024 May 6. Angiogenesis. 2024. PMID: 38709389 Free PMC article.
-
Functionalized screen-printed electrodes for non-invasive detection of vascular-endothelial cadherin in extracellular vesicles.RSC Adv. 2025 Apr 22;15(16):12609-12621. doi: 10.1039/d4ra08926j. eCollection 2025 Apr 16. RSC Adv. 2025. PMID: 40264865 Free PMC article.
References
-
- Antonetti DA, Klein R, Gardner TW. Diabetic retinopathy. N Engl J Med 2012;366(13): 1227–39. - PubMed
-
- Ghirlanda G, Di Leo MA, Caputo S, Cercone S, Greco AV. From functional to microvascular abnormalities in early diabetic retinopathy. Diabetes Metab Rev 1997;13(1):15–35. - PubMed
-
- Barber AJ, Baccouche B. Neurodegeneration in diabetic retinopathy: Potential for novel therapies. Vision Res 2017;139:82–92. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
