Non-invasive respiratory support in SARS-CoV-2 related acute respiratory distress syndrome: when is it most appropriate to start treatment?
- PMID: 36463178
- PMCID: PMC9719658
- DOI: 10.1186/s12931-022-02258-5
Non-invasive respiratory support in SARS-CoV-2 related acute respiratory distress syndrome: when is it most appropriate to start treatment?
Abstract
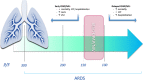
Background: Acute respiratory distress syndrome (ARDS) is one of the most severe complications of SARS-CoV-2 infection. Non-Invasive Respiratory Support (NRS) as Continuous Positive Airway Pressure (CPAP) and/or Non-Invasive Ventilation (NIV) has been proven as effective in the management of SARS-CoV-2-related ARDS. However, the most appropriate timing for start NRS is unknown.
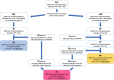
Methods: We conducted a prospective pilot study including all consecutive patients who developed moderate SARS-CoV-2-related ARDS during hospitalization. Patients were randomly divided into two intervention groups according to ARDS severity (assessed by PaO2/FiO2-P/F) at NRS beginning: group A started CPAP/NIV when P/F was ≤ 200 and group B started CPAP/NIV when P/F was ≤ 150. Eligible patients who did not give their consent to CPAP/NIV until the severe stage of ARDS and started non-invasive treatment when P/F ≤ 100 (group C) was added. The considered outcomes were in-hospital mortality, oro-tracheal intubation (OTI) and days of hospitalization.
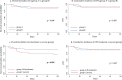
Results: Among 146 eligible patients, 29 underwent CPAP/NIV when P/F was ≤ 200 (Group A), 68 when P/F was ≤ 150 (Group B) and 31 patients agreed to non-invasive treatment only when P/F was ≤ 100 (Group C). Starting NRS at P/F level between 151 and 200 did not results in significant differences in the outcomes as compared to treatment starting with P/F ranging 101-150. Conversely, patients undergone CPAP/NIV in a moderate stage (P/F 101-200) had a significantly lower in-hospital mortality rate (13.4 vs. 29.0%, p = 0.044) and hospitalization length (14 vs. 15 days, p = 0.038) than those in the severe stage (P/F ≤ 100). Age and need for continuous ventilation were independent predictors of CPAP/NIV failure.
Conclusions: Starting CPAP/NIV in patients with SARS-CoV-2-related ARDS in moderate stage (100 > P/F ≤ 200) is associated to a reduction of both in-hospital mortality and hospitalization length compared to the severe stage (P/F ≤ 100). Starting CPAP/NIV with a P/F > 150 does not appear to be of clinical utility.
Keywords: ARDS; COVID-19; CPAP; NIV; SARS-CoV-2.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

References
-
- Grieco DL, Menga LS, Cesarano M, Rosà T, Spadaro S, Bitondo MM, COVID-ICU Gemelli Study Group et al. Effect of helmet noninvasive ventilation vs high-flow nasal oxygen on days free of respiratory support in patients with COVID-19 and moderate to severe hypoxemic respiratory failure: the HENIVOT randomized clinical trial. JAMA. 2021;325:1731–1743. doi: 10.1001/jama.2021.4682. - DOI - PMC - PubMed
-
- Bertaina M, Nuñez-Gil IJ, Franchin L, Fernández Rozas I, Arroyo-Espliguero R, Viana-Llamas MC, HOPE COVID-19 investigators et al. Non-invasive ventilation for SARS-CoV-2 acute respiratory failure: a subanalysis from the HOPE COVID-19 registry. Emerg Med J. 2021;38:359–365. doi: 10.1136/emermed-2020-210411. - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

