Transcutaneous vagal nerve stimulation to treat disorders of consciousness: Protocol for a double-blind randomized controlled trial
- PMID: 36467262
- PMCID: PMC9712558
- DOI: 10.1016/j.ijchp.2022.100360
Transcutaneous vagal nerve stimulation to treat disorders of consciousness: Protocol for a double-blind randomized controlled trial
Abstract
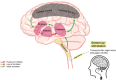
Background: Patients with disorders of consciousness (DoC) are a challenging population prone to misdiagnosis with limited effective treatment options. Among neuromodulation techniques, transcutaneous auricular vagal nerve stimulation (taVNS) may act through a bottom-up manner to modulate thalamo-cortical connectivity and promote patients' recovery. In this clinical trial, we aim to (1) assess the therapeutic clinical effects of taVNS in patients with DoC; (2) investigate the neural mechanisms underlying the effects of its action; (3) assess the feasibility and safety of the procedure in this challenging population; (4) define the phenotype of clinical responders; and (5) assess the long-term efficacy of taVNS in terms of functional outcomes.
Methods: We will conduct a prospective parallel randomized controlled double-blind clinical trial investigating the effects of taVNS as a treatment in DoC patients. Forty-four patients in the early period post-injury (7 to 90 days following the injury) will randomly receive 5 days of either active bilateral vagal stimulation (45 min duration with 30s alternative episodes of active/rest periods; 3mA; 200-300μs current width, 25Hz.) or sham stimulation. Behavioural (i.e., Coma Recovery Scale-Revised, CRS-R) and neurophysiological (i.e., high-density electroencephalography, hd-EEG) measures will be collected at baseline and at the end of the 5-day treatment. Analyses will seek for changes in the CRS-R and the EEG metrics (e.g., alpha band power spectrum, functional connectivity) at the group and individual (i.e., responders) levels.
Discussion: These results will allow us to investigate the vagal afferent network and will contribute towards a definition of the role of taVNS for the treatment of patients with DoC. We aim to identify the neural correlates of its action and pave the way to novel targeted therapeutic strategies.
Clinical trial registration: Clinicaltrials.gov n° NCT04065386.
Keywords: Behavior; Coma; Disorders of consciousness; Electroencephalography; Minimally conscious state; Randomized clinical trial; Treatment; Unresponsive wakefulness syndrome; Vagal nerve stimulation; Vegetative state.
© 2022 The Authors.
Conflict of interest statement
Authors have no conflict of interest to declare.
Figures

References
-
- Angelakis E., Konstantinidi M., Sakas D. Cognitive rehabilitation in disorders of consciousness with transcranial direct current stimulation. Dialogues in Clinical Neuroscience & Mental Health. 2018;1:1. doi: 10.26386/obrela.v1is3.54. - DOI
-
- Annen, J., Filippini, M.M., Bonin, E., Cassol, H., Aubinet, C., Carrière, M., Gosseries, O., Thibaut, A., Barra, A., Wolff, A., Sanz, L.R.D., Martial, C., Laureys, S., & Chatelle, C. (2019). Diagnostic accuracy of the CRS-R index in patients with disorders of consciousness. 10.1080/02699052.2019.1644376, 33(11), 1409–1412. 10.1080/02699052.2019.1644376 - PubMed
-
- Ardesch J.J., Buschman H.P.J., van der Burgh P.H., Wagener-Schimmel L.J.J.C., van der Aa H.E., Hageman G. Cardiac responses of vagus nerve stimulation: Intraoperative bradycardia and subsequent chronic stimulation. Clinical Neurology and Neurosurgery. 2007;109:849–852. doi: 10.1016/j.clineuro.2007.07.024. - DOI - PubMed
Associated data
LinkOut - more resources
Full Text Sources
Medical

