Efficacy and safety of COVID-19 vaccines
- PMID: 36473651
- PMCID: PMC9726273
- DOI: 10.1002/14651858.CD015477
Efficacy and safety of COVID-19 vaccines
Abstract
Background: Different forms of vaccines have been developed to prevent the SARS-CoV-2 virus and subsequent COVID-19 disease. Several are in widespread use globally. OBJECTIVES: To assess the efficacy and safety of COVID-19 vaccines (as a full primary vaccination series or a booster dose) against SARS-CoV-2.
Search methods: We searched the Cochrane COVID-19 Study Register and the COVID-19 L·OVE platform (last search date 5 November 2021). We also searched the WHO International Clinical Trials Registry Platform, regulatory agency websites, and Retraction Watch.
Selection criteria: We included randomized controlled trials (RCTs) comparing COVID-19 vaccines to placebo, no vaccine, other active vaccines, or other vaccine schedules.
Data collection and analysis: We used standard Cochrane methods. We used GRADE to assess the certainty of evidence for all except immunogenicity outcomes. We synthesized data for each vaccine separately and presented summary effect estimates with 95% confidence intervals (CIs). MAIN RESULTS: We included and analyzed 41 RCTs assessing 12 different vaccines, including homologous and heterologous vaccine schedules and the effect of booster doses. Thirty-two RCTs were multicentre and five were multinational. The sample sizes of RCTs were 60 to 44,325 participants. Participants were aged: 18 years or older in 36 RCTs; 12 years or older in one RCT; 12 to 17 years in two RCTs; and three to 17 years in two RCTs. Twenty-nine RCTs provided results for individuals aged over 60 years, and three RCTs included immunocompromized patients. No trials included pregnant women. Sixteen RCTs had two-month follow-up or less, 20 RCTs had two to six months, and five RCTs had greater than six to 12 months or less. Eighteen reports were based on preplanned interim analyses. Overall risk of bias was low for all outcomes in eight RCTs, while 33 had concerns for at least one outcome. We identified 343 registered RCTs with results not yet available. This abstract reports results for the critical outcomes of confirmed symptomatic COVID-19, severe and critical COVID-19, and serious adverse events only for the 10 WHO-approved vaccines. For remaining outcomes and vaccines, see main text. The evidence for mortality was generally sparse and of low or very low certainty for all WHO-approved vaccines, except AD26.COV2.S (Janssen), which probably reduces the risk of all-cause mortality (risk ratio (RR) 0.25, 95% CI 0.09 to 0.67; 1 RCT, 43,783 participants; high-certainty evidence). Confirmed symptomatic COVID-19 High-certainty evidence found that BNT162b2 (BioNtech/Fosun Pharma/Pfizer), mRNA-1273 (ModernaTx), ChAdOx1 (Oxford/AstraZeneca), Ad26.COV2.S, BBIBP-CorV (Sinopharm-Beijing), and BBV152 (Bharat Biotect) reduce the incidence of symptomatic COVID-19 compared to placebo (vaccine efficacy (VE): BNT162b2: 97.84%, 95% CI 44.25% to 99.92%; 2 RCTs, 44,077 participants; mRNA-1273: 93.20%, 95% CI 91.06% to 94.83%; 2 RCTs, 31,632 participants; ChAdOx1: 70.23%, 95% CI 62.10% to 76.62%; 2 RCTs, 43,390 participants; Ad26.COV2.S: 66.90%, 95% CI 59.10% to 73.40%; 1 RCT, 39,058 participants; BBIBP-CorV: 78.10%, 95% CI 64.80% to 86.30%; 1 RCT, 25,463 participants; BBV152: 77.80%, 95% CI 65.20% to 86.40%; 1 RCT, 16,973 participants). Moderate-certainty evidence found that NVX-CoV2373 (Novavax) probably reduces the incidence of symptomatic COVID-19 compared to placebo (VE 82.91%, 95% CI 50.49% to 94.10%; 3 RCTs, 42,175 participants). There is low-certainty evidence for CoronaVac (Sinovac) for this outcome (VE 69.81%, 95% CI 12.27% to 89.61%; 2 RCTs, 19,852 participants). Severe or critical COVID-19 High-certainty evidence found that BNT162b2, mRNA-1273, Ad26.COV2.S, and BBV152 result in a large reduction in incidence of severe or critical disease due to COVID-19 compared to placebo (VE: BNT162b2: 95.70%, 95% CI 73.90% to 99.90%; 1 RCT, 46,077 participants; mRNA-1273: 98.20%, 95% CI 92.80% to 99.60%; 1 RCT, 28,451 participants; AD26.COV2.S: 76.30%, 95% CI 57.90% to 87.50%; 1 RCT, 39,058 participants; BBV152: 93.40%, 95% CI 57.10% to 99.80%; 1 RCT, 16,976 participants). Moderate-certainty evidence found that NVX-CoV2373 probably reduces the incidence of severe or critical COVID-19 (VE 100.00%, 95% CI 86.99% to 100.00%; 1 RCT, 25,452 participants). Two trials reported high efficacy of CoronaVac for severe or critical disease with wide CIs, but these results could not be pooled. Serious adverse events (SAEs) mRNA-1273, ChAdOx1 (Oxford-AstraZeneca)/SII-ChAdOx1 (Serum Institute of India), Ad26.COV2.S, and BBV152 probably result in little or no difference in SAEs compared to placebo (RR: mRNA-1273: 0.92, 95% CI 0.78 to 1.08; 2 RCTs, 34,072 participants; ChAdOx1/SII-ChAdOx1: 0.88, 95% CI 0.72 to 1.07; 7 RCTs, 58,182 participants; Ad26.COV2.S: 0.92, 95% CI 0.69 to 1.22; 1 RCT, 43,783 participants); BBV152: 0.65, 95% CI 0.43 to 0.97; 1 RCT, 25,928 participants). In each of these, the likely absolute difference in effects was fewer than 5/1000 participants. Evidence for SAEs is uncertain for BNT162b2, CoronaVac, BBIBP-CorV, and NVX-CoV2373 compared to placebo (RR: BNT162b2: 1.30, 95% CI 0.55 to 3.07; 2 RCTs, 46,107 participants; CoronaVac: 0.97, 95% CI 0.62 to 1.51; 4 RCTs, 23,139 participants; BBIBP-CorV: 0.76, 95% CI 0.54 to 1.06; 1 RCT, 26,924 participants; NVX-CoV2373: 0.92, 95% CI 0.74 to 1.14; 4 RCTs, 38,802 participants). For the evaluation of heterologous schedules, booster doses, and efficacy against variants of concern, see main text of review.
Authors' conclusions: Compared to placebo, most vaccines reduce, or likely reduce, the proportion of participants with confirmed symptomatic COVID-19, and for some, there is high-certainty evidence that they reduce severe or critical disease. There is probably little or no difference between most vaccines and placebo for serious adverse events. Over 300 registered RCTs are evaluating the efficacy of COVID-19 vaccines, and this review is updated regularly on the COVID-NMA platform (covid-nma.com). Implications for practice Due to the trial exclusions, these results cannot be generalized to pregnant women, individuals with a history of SARS-CoV-2 infection, or immunocompromized people. Most trials had a short follow-up and were conducted before the emergence of variants of concern. Implications for research Future research should evaluate the long-term effect of vaccines, compare different vaccines and vaccine schedules, assess vaccine efficacy and safety in specific populations, and include outcomes such as preventing long COVID-19. Ongoing evaluation of vaccine efficacy and effectiveness against emerging variants of concern is also vital.
Copyright © 2022 The Authors. Cochrane Database of Systematic Reviews published by John Wiley & Sons, Ltd. on behalf of The Cochrane Collaboration.
Conflict of interest statement
Carolina Graña: none known.
Lina Ghosn: none known.
Theodoros Evrenoglou: none known.
Alexander Jarde: none known.
Silvia Minozzi: no relevant interests; Joint Co‐ordinating Editor and Method editor of the Drugs and Alcohol Group.
Hanna Bergman: Cochrane Response – consultant; WHO – grant/contract (Cochrane Response was commissioned by the WHO to perform review tasks that contribute to this publication).
Brian Buckley: none known.
Katrin Probyn: Cochrane Response – consultant; WHO – consultant (Cochrane Response was commissioned to perform review tasks that contribute to this publication).
Gemma Villanueva: Cochrane Response – employment (Cochrane Response has been commissioned by WHO to perform parts of this systematic review).
Nicholas Henschke: Cochrane Response – consultant; WHO – consultant (Cochrane Response was commissioned by the WHO to perform review tasks that contributed to this publication).
Hillary Bonnet: none known.
Rouba Assi: none known.
Sonia Menon: P95 – consultant.
Melanie Marti: no relevant interests; Medical Officer at WHO.
Declan Devane: Health Research Board (HRB) – grant/contract; registered nurse and registered midwife but no longer in clinical practice; Editor, Cochrane Pregnancy and Childbirth Group.
Patrick Mallon: AstraZeneca – Advisory Board; spoken of vaccine effectiveness to media (print, online, and live); works as a consultant in a hospital that provides vaccinations; employed by St Vincent's University Hospital.
Jean‐Daniel Lelievre: no relevant interests; published numerous interviews in the national press on the subject of COVID vaccination; Head of the Department of Infectious Diseases and Clinical Immunology CHU Henri Mondor APHP, Créteil; WHO (IVRI‐AC): expert Vaccelarate (European project on COVID19 Vaccine): head of WP; involved with COVICOMPARE P et M Studies (APHP, INSERM) (public fundings).
Lisa Askie: no relevant interests; Co‐convenor, Cochrane Prospective Meta‐analysis Methods Group.
Tamara Kredo: no relevant interests; Medical Officer in an Infectious Diseases Clinic at Tygerberg Hospital, Stellenbosch University.
Gabriel Ferrand: none known.
Mauricia Davidson: none known.
Carolina Riveros: no relevant interests; works as an epidemiologist.
David Tovey: no relevant interests; Emeritus Editor in Chief, Feedback Editors for 2 Cochrane review groups.
Joerg J Meerpohl: no relevant interests; member of the German Standing Vaccination Committee (STIKO).
Giacomo Grasselli: Pfizer – speaking engagement.
Gabriel Rada: none known.
Asbjørn Hróbjartsson: no relevant interests; Cochrane Methodology Review Group Editor.
Philippe Ravaud: no relevant interests; involved with Mariette CORIMUNO‐19 Collaborative 2021, the Ministry of Health, Programme Hospitalier de Recherche Clinique, Foundation for Medical Research, and AP‐HP Foundation.
Anna Chaimani: none known.
Isabelle Boutron: no relevant interests; member of Cochrane Editorial Board.
Figures

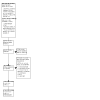






















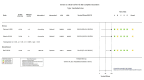





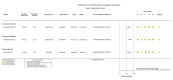







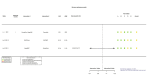

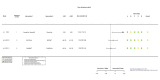





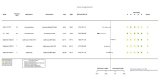
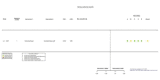
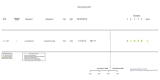




References
References to studies included in this review
Ali 2021 {published data only}
Al Kaabi 2021 {published data only}
Asano 2022 {published data only}
Bonelli 2021 {published data only}
-
- Bonelli M, Mrak D, Tobudic S, Sieghart D, Koblischke M, Mandl P, et al. Additional heterologous versus homologous booster vaccination in immunosuppressed patients without SARS-CoV-2 antibody seroconversion after primary mRNA vaccination: a randomized controlled trial. medRxiv 2021 [Preprint]. [DOI: 10.1101/2021.09.05.21263125] - DOI - PubMed
Bueno 2021 {published data only}
-
- Bueno SM, Abarca K, González PA, Gálvez NM, Soto JA, Duarte LF, et al. Interim report: safety and immunogenicity of an inactivated vaccine against SARS-CoV-2 in healthy Chilean adults in a phase 3 clinical trial. medRxiv 2021 [Preprint]. [DOI: 10.1101/2021.03.31.21254494] - DOI
Clemens 2021 {published data only}
Dunkle 2021 {published data only}
Ella 2021a {published data only}
Ella 2021b {published data only}
El Sahly 2021 {published data only}
-
- Food and Drug Administration. Vaccines and Related Biological Products Advisory Committee Meeting; December 17, 2020; FDA Briefing Document: Moderna COVID-19 vaccine. www.fda.gov/media/144434/download (accessed prior to 1 November 2022).
Emary 2021 {published data only}
Fadlyana 2021 {published data only}
-
- Fadlyana E, Rusmil K, Tarigan R, Rahmadi AR, Prodjosoewojo S, Sofiatin Y, et al. A phase III, observer-blind, randomized, placebo-controlled study of the efficacy, safety, and immunogenicity of SARS-CoV-2 inactivated vaccine in healthy adults aged 18–59 years: an interim analysis in Indonesia. Vaccine 2021;39(44):6520-8. - PMC - PubMed
Falsey 2021 {published data only}
Formica 2021 {published data only}
Frenck 2021 {published data only}
Guo 2021 {published data only}
Hall 2021 {published data only}
Han 2021 {published data only}
-
- Han B, Song Y, Li C, Yang W, Ma Q, Jiang Z, et al. Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine (CoronaVac) in healthy children and adolescents: a double-blind, randomized, controlled, phase 1/2 clinical trial. Lancet Infectious Diseases 2021;21(12):1645-53. - PMC - PubMed
Heath 2021 {published data only}
-
- Heath PT, Galiza EP, Baxter DN, Boffito M, Browne D, Burns F, et al . Efficacy of the NVX-CoV2373 Covid-19 vaccine against the B. 1.1.7 variant. medRxiv 2021 [Preprint].
Keech 2020 {published data only}
Kremsner 2021 {published data only}
-
- Kremsner PG, Ahuad Guerrero RA, Arana-Arri E, Aroca Martinez GJ, Bonten M, Chandler R, et al. Efficacy and safety of the CVnCoV SARS-CoV-2 mRNA vaccine candidate: results from Herald, a phase 2b/3, randomized, observer-blinded, placebo-controlled clinical trial in ten countries in Europe and Latin America. SSRN 2021 [Preprint]. [DOI: 10.2139/ssrn.3911826] - DOI - PMC - PubMed
Kulkarni 2021 {published data only}
Li 2021a {published data only}
-
- Li J, Hou L, Guo X, Jin P, Wu S, Zhu J, et al. Heterologous prime-boost immunization with CoronaVac and Convidecia. medRxiv 2021 [Preprint]. [DOI: 10.1101/2021.09.03.21263062] - DOI
Liu 2021 {published data only}
-
- Liu X, Shaw RH, Stuart AS, Greenland M, Aley PK, Andrews NJ, et al. Safety and immunogenicity of heterologous versus homologous prime-boost schedules with an adenoviral vectored and mRNA COVID-19 vaccine (Com-COV): a single-blind, randomized, non-inferiority trial. Lancet 2021;398(10303):856-69. - PMC - PubMed
-
- Liu X, Shaw RH, Stuart AS, Greenland M, Aley PK, Andrews NJ, et al. Safety and immunogenicity report from the Com-COV Study – a single-blind randomized non-inferiority trial comparing heterologous and homologous prime-boost schedules with an adenoviral vectored and mRNA COVID-19 vaccine. SSRN 2021 [Preprint]. [DOI: 10.2139/ssrn.3874014] - DOI - PMC - PubMed
Logunov 2021 {published data only}
-
- Logunov DY, Dolzhikova IV, Shcheblyakov DV, Tukhvatulin AI, Zubkova OV, Dzharullaeva AS, et al. Safety and efficacy of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine: an interim analysis of a randomized controlled phase 3 trial in Russia. Lancet 2021;397(10275):671-81. - PMC - PubMed
Madhi 2021a {published data only}
-
- Madhi SA, Koen AL, Izu A, Fairlie L, Cutland CL, Baillie V, et al. Safety and immunogenicity of the ChAdOx1 nCoV-19 (AZD1222) vaccine against SARS-CoV-2 in people living with and without HIV in South Africa: an interim analysis of a randomized, double-blind, placebo-controlled, phase 1B/2A trial. Lancet HIV 2021;8(9):e568-80. - PMC - PubMed
Madhi 2021b {published data only}
Mok 2021 {published data only}
-
- Mok C, Cheng S, Chen C, Yiu K, Chan TO, Lai KC, et al. A RCT of a third dose CoronaVac or BNT162b2 vaccine in adults with two doses of CoronaVac. medRxiv 2021 [Preprint]. [DOI: 10.1101/2021.11.02.21265843] - DOI
Palacios 2020 {published data only}
-
- Palacios R, Patiño EG, Oliveira Piorelli R, Conde MT, Batista AP, Zeng G, et al. Double-blind, randomized, placebo-controlled phase III clinical trial to evaluate the efficacy and safety of treating healthcare professionals with the adsorbed COVID-19 (inactivated) vaccine manufactured by Sinovac – PROFISCOV: a structured summary of a study protocol for a randomized controlled trial. Trials 2020;21(1):853. - PMC - PubMed
Sablerolles 2021 {published data only}
-
- Sablerolles RS, Rietdijk WJ, Goorhuis A, Postma DF, Visser LG, Geers D, et al. Immunogenicity and reactogenicity of booster vaccinations after Ad26.COV2.S priming. medRxiv 2021 [Preprint]. [DOI: 10.1101/2021.10.18.21264979] - DOI
Sadoff 2021a {published data only}
-
- Food and Drug Administration. Vaccines and related biological products Advisory Committee Meeting; February 26, 2021; FDA briefing document: Janssen Ad26.COV2.S vaccine for the prevention of COVID-19. www.fda.gov/media/146217/download (accessed prior to 1 November 2022).
-
- Sadoff J, Le Gars M, Shukarev G, Heerwegh D, Truyers C, Groot AM, et al. Safety and immunogenicity of the Ad26.COV2.S COVID-19 vaccine candidate: interim results of a phase 1/2a, double-blind, randomized, placebo-controlled trial. medRxiv 2020 [Preprint]. [DOI: 10.1101/2020.09.23.20199604] - DOI
Sadoff 2021b {published data only}
Shinde 2021 {published data only}
Tanriover 2021 {published data only}
Thomas 2021 {published data only}
-
- Food and Drug Administration. Vaccines and related biological products Advisory Committee Meeting; December 10, 2020; FDA briefing document: Pfizer-BioNTech COVID-19 vaccine. www.fda.gov/media/144245/download (accessed prior to 1 November 2022).
Toledo‐Romani 2021 {published data only}
-
- Toledo-Romani ME, Garcia-Carmenate M, Silva-Valenzuela C, Baldoquin-Rodriguez W, Martínez-Pérez M, Rodríguez-González M, et al. Safety and efficacy of the two doses conjugated protein-based SOBERANA-02 COVID-19 vaccine and of a heterologous three-dose combination with SOBERANA-PLUS: double-blind, randomized, placebo-controlled phase 3 clinical trial. medRxiv 2021 [Preprint]. [DOI: 10.1101/2021.10.31.21265703] - DOI - PMC - PubMed
Voysey 2021a {published data only}
-
- Voysey M, Clemens SA, Madhi SA, Weckx LY, Folegatti PM, Aley PK, et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomized controlled trials in Brazil, South Africa, and the UK. Lancet 2021;397(10269):99-111. [PMID: ] - PMC - PubMed
Walsh 2020 {published data only}
-
- Walsh EE, Frenck R, Falsey AR, Kitchin N, Absalon J, Gurtman A, et al. RNA-based COVID-19 vaccine BNT162b2 selected for a pivotal efficacy study. medRxiv 2020 [Preprint]. [DOI: 10.1101/2020.08.17.20176651] - DOI
Wu 2021a {published data only}
-
- Wu Z, Hu Y, Xu M, Chen Z, Yang W, Jiang Z, et al. Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine (CoronaVac) in healthy adults aged 60 years and older: a randomized, double-blind, placebo-controlled, phase 1/2 clinical trial. Lancet Infectious Diseases 2021;21(6):803-12. - PMC - PubMed
Xia 2020 {published data only}
Xia 2021 {published data only}
Zhang 2021 {published data only}
References to studies excluded from this review
Baden 2021 {published data only}
Barrett 2021 {published data only}
-
- Barrett JR, Belij-Rammerstorfer S, Dold C, Ewer KJ, Folegatti PM, Gilbride C, et al. Phase 1/2 trial of SARS-CoV-2 vaccine ChAdOx1 nCoV-19 with a booster dose induces multifunctional antibody responses. Nature Medicine 2021;27(2):279-88. - PubMed
Ewer 2021 {published data only}
-
- Ewer KJ, Barrett JR, Belij-Rammerstorfer S, Sharpe H, Makinson R, Morter R, et al. T cell and antibody responses induced by a single dose of ChAdOx1 nCoV-19 (AZD1222) vaccine in a phase 1/2 clinical trial. Nature Medicine 2021;27(2):270-8. - PubMed
Flaxman 2021 {published data only}
Hsieh 2021 {published data only}
-
- Hsieh SM, Liu WD, Huang YS, Lin YJ, Hsieh EF, Lian WC, et al. Safety and immunogenicity of a recombinant stabilized prefusion SARS-CoV-2 spike protein vaccine (MVC-COV1901) adjuvanted with CpG 1018 and aluminum hydroxide in healthy adults: a phase 1, dose-escalation study. eClinicalMedicine 2021;38:100989. - PMC - PubMed
Irfan 2021 {published data only}
-
- Irfan N, Chagla Z. In South Africa, a 2-dose Oxford/AZ vaccine did not prevent mild to moderate COVID-19 (cases mainly B.1.351 variant). Annals of Internal Medicine 2021;174(5):JC50. - PubMed
Lazarus 2021 {published data only}
-
- Lazarus R, Baos S, Cappel-Porter H, Carson-Stevens A, Clout M, Culliford L, et al. The safety and immunogenicity of concomitant administration of COVID-19 vaccines (ChAdOx1 or BNT162b2) with seasonal influenza vaccines in adults: a phase IV, multicentre randomized controlled trial with blinding (ComFluCOV). SSRN 2021 [Preprint]. [DOI: 10.2139/ssrn.3931758] - DOI - PMC - PubMed
Patamatamkul 2021 {published data only}
-
- Patamatamkul S, Thammawat S, Buranrat B. Induction of robust neutralizing antibodies against the COVID-19 Delta variant with ChAdOx1 nCoV-19 or BNT162b2 as a booster following a primary vaccination series with CoronaVac. medRxiv 2021 [Preprint]. [DOI: 10.1101/2021.09.25.21264099] - DOI
Ward 2021a {published data only}
-
- Ward BJ, Séguin A, Couillard J, Trépanier S, Landry N. Phase III: randomized observer-blind trial to evaluate lot-to-lot consistency of a new plant-derived quadrivalent virus like particle influenza vaccine in adults 18-49 years of age. Vaccine 2021;39(10):1528-33. - PubMed
Wu 2021b {published data only}
Additional references
Abbasi 2020
-
- Abbasi J. COVID-19 and mRNA vaccines – first large test for a new approach. JAMA 2020;324(12):1125-7. - PubMed
Angkasekwinai 2022
-
- Angkasekwinai N, Sewatanon J, Niyomnaitham S, Phumiamorn S, Sukapirom K, Sapsutthipas S, et al. Comparison of safety and immunogenicity of CoronaVac and ChAdOx1 against the SARS-CoV-2 circulating variants of concern (Alpha, Delta, Beta) in Thai healthcare workers. Vaccine X 2022;10:100153. - PMC - PubMed
Attaway 2021
-
- Attaway AH, Scheraga RG, Bhimraj A, Biehl M, Hatipoğlu U. Severe Covid-19 pneumonia: pathogenesis and clinical management. BMJ 2021;372:n436. - PubMed
Baden 2021
Balduzzi 2019
Borobia 2021
Boutron 2020a
Boutron 2020b
-
- Boutron I, Chaimani A, Meerpohl JJ, Hróbjartsson A, Devane D, Rada G, et al. Interventions for preventing and treating COVID-19: protocol for a living mapping of research and a living systematic review. Cochrane Database of Systematic Reviews 2020, Issue 11. Art. No: CD013769. [DOI: 10.1002/14651858.CD013769] - DOI
Bucci 2021
Bueno 2021
-
- Bueno SM, Abarca K, González PA, Gálvez NM, Soto JA, Duarte LF, et al. Interim report: safety and immunogenicity of an inactivated vaccine against SARS-CoV-2 in healthy Chilean adults in a phase 3 clinical trial. medRxiv 2021 [Preprint].
Cabanac 2021
Castagneto Gissey 2021
-
- Castagneto Gissey L, Panunzi S, Maltese S, Russo MF, Angelini G, De Gaetano A, et al. Living systematic meta-analysis of COVID-19 vaccines and dose allocation strategies. SSRN 2021 [Preprint]. [DOI: 10.2139/ssrn.3827806] - DOI
CDC 2021
-
- Centers for Disease Control and Prevention. Understanding mRNA COVID-19 vaccines. www.cdc.gov/coronavirus/2019-ncov/vaccines/different-vaccines/mrna.html (accessed prior to 1 November 2022).
Chaimani 2013
Chaimani 2015
-
- Chaimani A, Salanti G. Visualizing assumptions and results in network meta-analysis: the Network Graphs Package. Stata Journal 2015;15(4):905-50.
Chaimani 2022
-
- Chaimani A, Caldwell DM, Li T, Higgins JP, Salanti G. Chapter 11: Undertaking network meta-analyses. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 6.3 (updated February 2022). Cochrane, 2022. Available from training.cochrane.org/handbook.
Chappell 2021
-
- Chappell KJ, Mordant FL, Li Z, Wijesundara DK, Ellenberg P, Lackenby JA, et al. Safety and immunogenicity of an MF59-adjuvanted spike glycoprotein-clamp vaccine for SARS-CoV-2: a randomised, double-blind, placebo-controlled, phase 1 trial. Lancet Infectious Diseases 2021;21(10):1383-94. - PMC - PubMed
Che 2021
Chu 2021
Dal‐Ré 2021a
Dal‐Ré 2021b
-
- Dal-Ré R, Orenstein W, Caplan AL. Being fair to participants in placebo-controlled COVID-19 vaccine trials. Nature Medicine 2021;27(6):938. - PubMed
DeZure 2016
Dias 2010
-
- Dias S, Welton NJ, Caldwell DM, Ades AE. Checking consistency in mixed treatment comparison meta-analysis. Statistics in Medicine 2010;29(7-8):932-44. - PubMed
Dong 2020
Egger 1997
Ella 2020b
-
- Ella R, Reddy S, Jogdand H, Sarangi V, Ganneru B, Prasad S, et al. Safety and immunogenicity clinical trial of an inactivated SARS-CoV-2 vaccine, BBV152 (a phase 2, double-blind, randomised controlled trial) and the persistence of immune responses from a phase 1 follow-up report. medRxiv 2020 [Preprint]. [DOI: 10.1101/2020.12.21.20248643] - DOI
Ella 2021a
-
- Ella R, Reddy S, Jogdand H, Sarangi V, Ganneru B, Prasad S, et al. Safety and immunogenicity of an inactivated SARS-CoV-2 vaccine, BBV152: interim results from a double-blind, randomized, multicentre, phase 2 trial, and 3-month follow-up of a double-blind, randomized phase 1 trial. Lancet Infectious Diseases 2021;21(7):950-61. - PMC - PubMed
Enjuanes 2016
Epistemonikos
-
- Epistemonikos. Epistemonikos L·OVE COVID-19 platform. Available at app.iloveevidence.com/loves/5e6fdb9669c00e4ac072701d?utm=ile.
FDA 2020a
-
- Food and Drug Administration. Development and licensure of vaccines to prevent COVID-19. Guidance for industry; June 2020. www.fda.gov/media/139638/download (accessed prior to 1 November 2022).
FDA 2020b
-
- Food and Drug Administration. FDA Briefing Document Moderna COVID-19 Vaccine. Vaccines and Related Biological Products Advisory Committee Meeting December 17, 2020. www.fda.gov/media/144434/download (accessed prior to 1 November 2022).
FDA 2020c
-
- Food and Drug Administration. FDA Briefing Document Pfizer-BioNTech COVID-19 Vaccine. Vaccines and Related Biological Products Advisory Committee Meeting December 10, 2020. www.fda.gov/media/144245/download (accessed prior to 1 November 2022).
FDA 2021
-
- Food and Drug Administration. FDA Briefing Document Janssen Ad26.COV2.S Vaccine for the Prevention of COVID-19. Vaccines and Related Biological Products Advisory Committee Meeting February 26, 2021. www.fda.gov/media/146217/download (accessed prior to 1 November 2022).
Feikin 2022
Feng 2021
Folegatti 2020
Formica 2021
Fuenmayor 2017
Gavi 2020
-
- Gavi. What are viral vector-based vaccines and how could they be used against COVID-19? www.gavi.org/vaccineswork/what-are-viral-vector-based-vaccines-and-how-c... (accessed prior to 1 November 2022).
Gobeil 2021
-
- Gobeil P, Pillet S, Séguin A, Boulay I, Mahmood A, Vinh DC, et al. Interim report of a Phase 2 randomized trial of a plant-produced virus-like particle vaccine for Covid-19 in healthy adults aged 18-64 and older adults aged 65 and older. medRxiv 2021 [Preprint]. [DOI: 10.1101/2021.05.14.21257248] - DOI
Goepfert 2021
-
- Goepfert PA, Fu B, Chabanon AL, Bonaparte MI, Davis MG, Essink BJ, et al. Safety and immunogenicity of SARS-CoV-2 recombinant protein vaccine formulations in healthy adults: interim results of a randomised, placebo-controlled, phase 1-2, dose-ranging study. Lancet Infectious Diseases 2021;21(9):1257-70. - PMC - PubMed
GRADEpro GDT [Computer program]
-
- GRADEpro GDT. Version accessed 6 December 2021. Hamilton (ON): McMaster University (developed by Evidence Prime). Available at gradepro.org.
Guyatt 2011
-
- Guyatt GH, Oxman AD, Kunz R, Brozek J, Alonso-Coello P, Rind D, et al. GRADE guidelines 6. Rating the quality of evidence – imprecision. Journal of Clinical Epidemiology 2011;64(12):1283-93. - PubMed
Harder 2021
Hayawi 2021
Heath 2021
-
- Heath PT, Galiza EP, Baxter DN, Boffito M, Browne D, Burns F, et al. Efficacy of the NVX-CoV2373 Covid-19 vaccine against the B.1.1.7 variant. medRxiv 2021 [Preprint]. [DOI: 10.1101/2021.05.13.21256639] - DOI
Higdon 2021
-
- Higdon MM, Wahl B, Jones CB, Rosen JG, Truelove SA, Baidya A, et al. A systematic review of COVID-19 vaccine efficacy and effectiveness against SARS-CoV-2 infection and disease. medRxiv 2021 [Preprint]. [DOI: 10.1101/2021.09.17.21263549] - DOI
Higgins 2021
-
- Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 6.2 (updated February 2021). Cochrane, 2021. Available from training.cochrane.org/handbook/archive/v6.2.
Hobernik 2018
Hultcrantz 2017
Kirkham 2018
Korang 2022
Kow 2022
Lazarus 2021
-
- Lazarus R, Taucher C, Brown C, Čorbic I, Danon L, Dubischar K, et al. Immunogenicity and safety of inactivated whole virion Coronavirus vaccine with CpG (VLA2001) in healthy adults aged 18 to 55: a randomised phase 1 /2 clinical trial. medRxiv 2021 [Preprint]. [DOI: 10.1101/2021.08.13.21262021] - DOI
Li 2021b
-
- Li J, Hui A, Zhang X, Yang Y, Tang R, Ye H, et al. Safety and immunogenicity of the SARS-CoV-2 BNT162b1 mRNA vaccine in younger and older Chinese adults: a randomized, placebo-controlled, double-blind phase 1 study. Nature Medicine 2021;27(6):1062-70. - PubMed
Li 2021c
-
- Li M, Yang J, Wang L, Wu Q, Wu Z, Zheng W, et al. A booster dose is immunogenic and will be needed for older adults who have completed two doses vaccination with CoronaVac: a randomized, double-blind, placebo-controlled, phase 1/2 clinical trial. medRxiv 2021 [Preprint]. [DOI: 10.1101/2021.08.03.21261544] - DOI
Liu 2021
-
- Liu X, Shaw RH, Stuart AS, Greenland M, Dinesh T, Provstgaard-Morys S, et al. Safety and immunogenicity report from the com-COV study – a single-blind randomised non-inferiority trial comparing heterologous and homologous prime-boost schedules with an adenoviral vectored and mRNA COVID-19 vaccine. SSRN 2021 [Preprint]. - PMC - PubMed
Low 2021
-
- Low JG, Alwis R, Chen S, Kalimuddin S, Leong YA, Mah TK, et al. A phase 1/2 randomized, double-blinded, placebo controlled ascending dose trial to assess the safety, tolerability and immunogenicity of ARCT-021 in healthy adults. medRxiv 2021 [Preprint]. [DOI: 10.1101/2021.07.01.21259831] - DOI
Lv 2021
Madhi 2021
Makris 2021
Mammen 2021
-
- Mammen MP Jr, Tebas P, Agnes J, Giffear M, Kraynyak KA, Blackwood E, et al. Safety and immunogenicity of INO-4800 DNA vaccine against SARS-CoV-2: a preliminary report of a randomized, blinded, placebo-controlled, Phase 2 clinical trial in adults at high risk of viral exposure. medRxiv 2021 [Preprint]. [DOI: 10.1101/2021.05.07.21256652] - DOI
Marshall 2020
Mavridis 2014
-
- Mavridis D, Welton NJ, Sutton A, Salanti G. A selection model for accounting for publication bias in a full network meta-analysis. Statistics in Medicine 2014;33(30):5399-412. - PubMed
Meng 2021b
-
- Meng FY, Gao F, Jia SY, Wu XH, Li JX, Guo XL, et al. Safety and immunogenicity of a recombinant COVID-19 vaccine (Sf9 cells) in healthy population aged 18 years or older: two single-center, randomised, double-blind, placebo-controlled, phase 1 and phase 2 trials. Signal Transduction and Targeted Therapy 2021;6(1):271. - PMC - PubMed
Mortola 2004
Mulligan 2020
-
- Mulligan MJ, Lyke KE, Kitchin N, Absalon J, Gurtman A, Lockhart S, et al. Phase I/II study of COVID-19 RNA vaccine BNT162b1 in adults. Nature 2020;586(7830):589-93. - PubMed
Nguyen 2021
Nikolakopoulou 2020
Oikonomidi 2020
Ostrowski 2021
Ouzzani 2016
Oxford Vaccine Group 2020
-
- Oxford Vaccine Group. Vaccine Knowledge Project: independent information about vaccines and infectious diseases. vk.ovg.ox.ac.uk/vk (accessed prior to 1 November 2022).
Page 2021
Pajon 2021
Pan 2021a
-
- Pan HX, Liu JK, Huang BY, Li GF, Chang XY, Liu YF, et al. Immunogenicity and safety of a SARS-CoV-2 inactivated vaccine (KCONVAC) in healthy adults: two randomized, double-blind, and placebo-controlled Phase 1/2 clinical trials. medRxiv 2021 [Preprint]. [DOI: 10.1101/2021.04.07.21253850] - DOI
Pan 2021b
-
- Pan HX, Wu QH, Zeng G, Yang J, Jiang DY, Deng XW, et al. Immunogenicity and safety of a third dose, and immune persistence of CoronaVac vaccine in healthy adults aged 18–59 years: interim results from a double-blind, randomized, placebo-controlled phase 2 clinical trial. medRxiv 2021 [Preprint]. [DOI: 10.1101/2021.07.23.21261026] - DOI
Pérez‐Rodríguez 2021
-
- Pérez-Rodríguez S, la Caridad Rodríguez-González M, Ochoa-Azze R, Climent-Ruiz Y, Alberto González-Delgado C, Paredes-Moreno B, et al. A randomized, double-blind phase I clinical trial of two recombinant dimeric RBD COVID-19 vaccine candidates: safety, reactogenicity and immunogenicity. medRxiv 2021 [Preprint]. [DOI: 10.1101/2021.10.04.21264522] - DOI - PMC - PubMed
Pitisuttithum 2021
-
- Pitisuttithum P, Luvira V, Lawpoolsri S, Muangnoicharoen S, Kamolratanakul S, Sivakorn C, et al. Safety and immunogenicity of an inactivated recombinant Newcastle disease virus vaccine expressing SARS-CoV-2 spike: interim results of a randomised, placebo-controlled, Phase 1/2 trial. medRxiv 2021 [Preprint]. [DOI: 10.1101/2021.09.17.21263758] - DOI - PMC - PubMed
Polack 2020
Pollard 2021
Pu 2021
Qiao 2020
Ramasamy 2020
-
- Ramasamy MN, Minassian AM, Ewer KJ, Flaxman AL, Folegatti PM, Owens DR, et al. Safety and immunogenicity of ChAdOx1 nCoV-19 vaccine administered in a prime-boost regimen in young and old adults (COV002): a single-blind, randomized, controlled, phase 2/3 trial. Lancet 2020;396(10267):1979-93. - PMC - PubMed
Richmond 2021
Riley 2011
-
- Riley RD, Higgins JP, Deeks JJ. Interpretation of random effects meta-analyses. BMJ 2011;342:d549. - PubMed
Rizk 2021
-
- Rizk JG, Gupta A, Sardar P, Henry BM, Lewin JC, Lippi G, et al. Clinical characteristics and pharmacological management of COVID-19 vaccine-induced immune thrombotic thrombocytopenia with cerebral venous sinus thrombosis: a review. JAMA Cardiology 2021;6(12):1451-60. - PubMed
Roozen 2021
-
- Roozen GV, Prins ML, Binnendijk R, den Hartog G, Kuiper VP, Prins C, et al. Tolerability, safety and immunogenicity of intradermal delivery of a fractional dose mRNA-1273 SARS-CoV-2 vaccine in healthy adults as a dose sparing strategy. medRxiv 2021 [Preprint]. [DOI: 10.1101/2021.07.27.21261116] - DOI
Roper 2009
Rotshild 2021
Rubin 2013
-
- Rubin LG, Levin MJ, Ljungman P, Davies EG, Avery R, Tomblyn M, et al. 2013 IDSA clinical practice guideline for vaccination of the immunocompromized host. Clinical Infectious Diseases 2013;58(3):e44-e100. - PubMed
Rucker 2013
-
- Rucker G, Schwarzer G, Rücker G, Schwarzer G, Krahn U, König J. Network meta-analysis using frequentist methods – package netmeta. cran.r–project.org (accessed prior to 1 November 2022).
Ryzhikov 2021
-
- Ryzhikov AB, Ryzhikov ЕА, Bogryantseva MP, Usova SV, Danilenko ED, Nechaeva EA, et al. A single blind, placebo-controlled randomized study of the safety, reactogenicity and immunogenicity of the "EpiVacCorona" vaccine for the prevention of COVID-19. Russian Journal of Infection and Immunity 2021;11(2):283-96.
Sadoff 2020c
-
- Sadoff J, Le Gars M, Shukarev G, Heerwegh D, Truyers C, Groot AM, et al. Safety and immunogenicity of the Ad26. COV2. S COVID-19 vaccine candidate: interim results of a phase 1/2a, double-blind, randomized, placebo-controlled trial. MedRxiv 2020 [Preprint].
Salanti 2011
-
- Salanti G, Ades AE, Ioannidis JP. Graphical methods and numerical summaries for presenting results from multiple-treatment meta-analysis: an overview and tutorial. Journal of Clinical Epidemiology 2011;64(2):163-7. - PubMed
Schünemann 2021
-
- Schünemann HJ, Higgins JP, Vist GE, Glasziou P, Akl EA, Skoetz N, et al. Chapter 14: Completing 'Summary of findings' tables and grading the certainty of the evidence. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 6.2 (updated February 2021). Cochrane, 2021. Available from training.cochrane.org/handbook/archive/v6.2.
Sharifian‐Dorche 2021
Shinde 2021
Shu 2021
-
- Shu YJ, He JF, Pei RJ, He P, Huang ZH, Chen SM, et al. Immunogenicity and safety of a recombinant fusion protein vaccine (V-01) against coronavirus disease 2019 in healthy adults: a randomized, double-blind, placebo-controlled, phase II trial. Chinese Medical Journal 2021;134(16):1967-76. - PMC - PubMed
Sridhar 2021
-
- Sridhar S, Joaquin A, Bonaparte MI, Bueso A, Chabanon AL, Chen A, et al. Safety and immunogenicity of a SARS-CoV-2 recombinant protein vaccine with AS03 adjuvant in healthy adults: interim findings from a phase 2, randomised, dose-finding, multi-centre study. medRxiv 2021 [Preprint]. [DOI: 10.1101/2021.10.08.21264302] - DOI - PMC - PubMed
Stephenson 2021
Sterne 2019
-
- Sterne JA, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: a revised tool for assessing risk of bias in randomized trials. BMJ 2019;366:l4898. - PubMed
Thomas 2021
Turner 2012
van Riel 2020
-
- Riel D, Wit E. Next-generation vaccine platforms for COVID-19. Nature Materials 2020;19(8):810-2. - PubMed
Viechtbauer 2010
-
- Viechtbauer W. Conducting meta-analyses in R with the metafor package. Journal of Statistical Software 2010;36(3):1-48.
Voysey 2021b
-
- Voysey M, Costa Clemens SA, Madhi SA, Weckx LY, Folegatti PM, Aley PK, et al. Single-dose administration and the influence of the timing of the booster dose on immunogenicity and efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine: a pooled analysis of four randomized trials. Lancet 2021;397(10277):881-91. - PMC - PubMed
Walsh 2021
-
- Walsh EE, Frenck R, Falsey AR, Kitchin N, Absalon J, Gurtman A, et al. RNA-based COVID-19 vaccine BNT162b2 selected for a pivotal efficacy study. medRxiv 2021 [Preprint].
Ward 2021b
White 2008
-
- White IR, Higgins JP, Wood AM. Allowing for uncertainty due to missing data in meta-analysis – part 1: two-stage methods. Statistics in Medicine 2008;27(5):711-27. - PubMed
WHO 2020a
-
- World Health Organization. Coronavirus disease 2019 (COVID-19). Situation report – 51. apps.who.int/iris/handle/10665/331475 (accessed prior to 1 November 2022).
WHO 2020b
-
- World Health Organization. Considerations for evaluation of COVID19 vaccines. Points to consider for manufacturers of COVID-19 vaccines. Version 24 September 2020. www.who.int/docs/default-source/in-vitro-diagnostics/covid19/who-evaluat... (accessed prior to 1 November 2022).
WHO 2020c
-
- World Health Organization. WHO target product profiles for COVID-19 vaccines. Version 3 – 29 April 2020. www.who.int/docs/default-source/blue-print/who-target-product-profiles-f... (accessed prior to 1 November 2022).
WHO 2022a
-
- World Health Organization. Tracking SARS-CoV-2 variants. www.who.int/activities/tracking-SARS-CoV-2-variants (accessed prior to 1 November 2022).
WHO 2022b
-
- World Health Organization. COVID-19 vaccine tracker and landscape. www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-va... (accessed prior to 1 November 2022).
WHO Ad Hoc Expert Group 2021
Worldometer 2022
-
- Worldometer. COVID-19 coronavirus pandemic. www.worldometers.info/coronavirus/#countries (accessed prior to 1 November 2022).
Wu 2021c
-
- Wu S, Huang J, Zhang Z, Wu J, Zhang J, Hu H, et al. Safety, tolerability, and immunogenicity of an aerosolised adenovirus type-5 vector-based COVID-19 vaccine (Ad5-nCoV) in adults: preliminary report of an open-label and randomised phase 1 clinical trial. Lancet Infectious Diseases 2021;21(12):1654-64. - PMC - PubMed
Xia 2020
Yang 2021
-
- Yang S, Li Y, Dai L, Wang J, He P, Li C, et al. Safety and immunogenicity of a recombinant tandem-repeat dimeric RBD-based protein subunit vaccine (ZF2001) against COVID-19 in adults: two randomized, double-blind, placebo-controlled, phase 1 and 2 trials. Lancet Infectious Diseases 2021;21(8):1107-19. - PMC - PubMed
Yepes‐Nuñez 2019
-
- Yepes-Nuñez JJ, Li SA, Guyatt G, Jack SM, Brozek JL, Beyene J, et al. Development of the summary of findings table for network meta-analysis. Journal of Clinical Epidemiology 2019;115:1-13. - PubMed
Zakarya 2021
Zeng 2021a
-
- Zeng L, Brignardello-Petersen R, Hultcrantz M, Siemieniuk RA, Santesso N, Traversy G, et al. GRADE guidelines 32: GRADE offers guidance on choosing targets of GRADE certainty of evidence ratings. Journal of Clinical Epidemiology 2021;137:163-75. - PubMed
Zeng 2021b
Zhang 2021b
Zhu 2020
Zhu 2021a
-
- Zhu F, Jin P, Zhu T, Wang W, Ye H, Pan H, et al. Safety and immunogenicity of a recombinant adenovirus type-5-vectored COVID-19 vaccine with a homologous prime-boost regimen in healthy participants aged 6 years and above: a randomized, double-blind, placebo-controlled, phase 2b trial. Clinical Infectious Diseases 2022;75(1):e783-91. [DOI: 10.1093/cid/ciab845] - DOI - PMC - PubMed
Zhu 2022
-
- Zhu F, Jin P, Zhu T, Wang W, Ye H, Pan H, et al. Safety and immunogenicity of a recombinant adenovirus type-5-vectored COVID-19 vaccine with a homologous prime-boost regimen in healthy participants aged 6 years and above: a randomized, double-blind, placebo-controlled, phase 2b trial. Clinical Infectious Diseases 2022;75(1):e783-91. - PMC - PubMed
References to other published versions of this review
Grana 2021
-
- Grana C, Ghosn L, Boutron I. Efficacy and safety of COVID-19 vaccines: a systematic review and meta-analysis. PROSPERO 2021 CRD42021271897. www.crd.york.ac.uk/prospero/display_record.php?RecordID=271897 (accessed prior to 1 November 2022).
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

