Comparison of dynamic changes in the peripheral CD8+ T cells function and differentiation in ESCC patients treated with radiotherapy combined with anti-PD-1 antibody or concurrent chemoradiotherapy
- PMID: 36479110
- PMCID: PMC9720318
- DOI: 10.3389/fimmu.2022.1060695
Comparison of dynamic changes in the peripheral CD8+ T cells function and differentiation in ESCC patients treated with radiotherapy combined with anti-PD-1 antibody or concurrent chemoradiotherapy
Abstract
Objective: The systematic immune status of cancer patients undergoing immunotherapy is little known. We prospectively identified the function and differentiation traits of peripheral CD8+ T cells based on our phase 1b clinical trial (NCT03222440) of radiotherapy combined with camrelizumab in patients with locally advanced esophageal squamous cell carcinoma (ESCC) and compared it with concurrent chemoradiotherapy (CCRT).
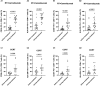
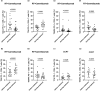
Methods: 19 and 18 patients were included in the cohort of radiotherapy plus camrelizumab and cohort of CCRT treatment. By using flow cytometry, we evaluated the expression levels of PD-1, Eomes, T-bet and IFN-γ (function), CD38 and HLA-DR (activation), and differentiation subsets classified according to the expression levels of CD45RA and CD62L in peripheral CD8+ T cells before and during treatment.
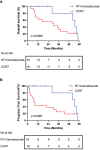
Results: Effective binding of anti-PD-1 antibody camrelizumab with PD-1 on CD8+ T cells was detected during treatment. Both two treatments elevated the expression levels of activation molecules CD38 and HLA-DR on CD8+ T cells. PD-1+CD8+ T cells had more activation features than PD-1-CD8+ T cells in two groups and the treatments did not alter these differences. The two treatments activated both PD-1+ and PD-1- CD8+ T cells. PD-1+CD8+ T cells had less Naïve and TEMRA but more Tcm and Tem than PD-1-CD8+ T cells in two groups and both two treatments changed the ratio of memory T cells in PD-1+ and PD-1- cells. RT plus camrelizumab treatment reduced Naïve T cells and TEMRA subsets both in PD-1+ and PD-1- CD8+ T cells while elevated Tcm subset in PD-1+CD8+ T cells and Tem subset in PD-1-CD8+ T cells. CCRT elevated Tcm subset and reduced TEMRA subset in PD-1-CD8+ T cells while did not change any subset in PD-1+CD8+ T cells. Furthermore, patients undergoing radiotherapy plus immunotherapy were found to obtain better prognosis than those receiving CCRT.
Conclusions: This study identified the dynamic changes of systematic immune status of patients undergoing treatment. The two treatments had similar activation effects on peripheral CD8+ T cells with different PD-1 properties but had different effects on their differentiation status. These results provided potential clues to the reasons underlying the difference in prognosis of the two treatments.
Keywords: CD8+ T cells; ESCC; PD-1; chemoradiotherapy; immunotherapy; radiotherapy.
Copyright © 2022 Wei, Li, Guo, Ma, Li, Wei, Han, Zhang, Chen, Yan, Zhou, Pang, Wang and Zhang.
Conflict of interest statement
Author JZ is employed by Jiangsu Hengrui Pharmaceuticals Co., Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



References
-
- Brahmer JR, Drake CG, Wollner I, Powderly JD, Picus J, Sharfman WH, et al. Phase I study of single-agent anti-programmed death-1 (MDX-1106) in refractory solid tumors: safety, clinical activity, pharmacodynamics, and immunologic correlates. J Clin Oncol (2010) 28(19):3167–75. doi: 10.1200/JCO.2009.26.7609 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials

