Understanding the effects of opioids vs non-opioids in the treatment of neonatal abstinence syndrome, an in vitro model
- PMID: 36483468
- PMCID: PMC9723166
- DOI: 10.3389/fped.2022.1068330
Understanding the effects of opioids vs non-opioids in the treatment of neonatal abstinence syndrome, an in vitro model
Abstract
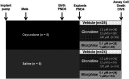
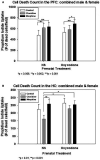
Neonatal abstinence syndrome (NAS) refers to cadre of withdrawal manifestations in infants born to mothers who used illicit and licit substances during pregnancy. The increasing prevalence of NAS has been largely due to the maternal use of opioids during pregnancy. NAS contributes to increased morbidity and long-term disability in surviving infants. Clinically, oral opioid therapies for opioid exposure have been a standard treatment with morphine (MO) being the most commonly used medication. Recently, a non-opioid agent, clonidine (CD) has also been used with potentially favorable short- and long-term outcomes in infants. However, data regarding the cellular and molecular effects of these treatments on the developing brain is still lacking due to a lack of a reliable animal model that targets the neonatal brain. To address this gap in knowledge we determined the effects of MO or CD on the cell death of neonatal cortical explant cultures that were exposed to oxycodone (OXY) in utero. Sprague Dawley rats were randomized and implanted with programmable infusion pumps before mating to receive either the OXY (dose increasing from 1.21-1.90 mg/kg/day to a maximum dose of 2.86-3.49 mg/kg/day) or normal saline (NS) throughout pregnancy and until one week after delivery. Male and female rat pups were sacrificed on postnatal day 4, and the prefrontal cortex (PFC) and hippocampus (HC) were dissected and treated with MO (0.10-1.00 µM) or CD (1.20-120.00 µM) in culture media. After 5 days of treatment the explants were labeled with propidium iodide to detect cell death. Dead cells were analyzed and counted under fluorescence microscopy. In explants from the PFC, cell death was greater in those prenatally exposed to OXY and postnatally treated with MO (OXY/MO) (736.8 ± 76.5) compared to OXY/CD (620.9 ± 75.0; p = 0.005). In the HC explants, mean cell death counts were not significantly different between groups regardless of prenatal exposure or postnatal treatment (p = 0.19). The PFC is vital in controlling higher-order executive functions such as behavioral flexibility, learning and working memory. Therefore, our finding is consistent with executive function problems in children with prenatal opioid exposure.
Keywords: cell death; clonidine; hippocampus; in vitro model; morphine; neonatal abstinence syndrome (NAS); opioid withdrawal; prefrontal cortex.
© 2022 Sithisarn, Legan, Westgate, Bada and Wilson.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Sedatives for opioid withdrawal in newborn infants.Cochrane Database Syst Rev. 2021 May 18;5(5):CD002053. doi: 10.1002/14651858.CD002053.pub4. Cochrane Database Syst Rev. 2021. PMID: 34002380 Free PMC article.
-
Postnatal oxytocin treatment improves survival and neurodevelopmental outcomes in an animal model of neonatal abstinence syndrome.Compr Psychoneuroendocrinol. 2022 May 18;11:100143. doi: 10.1016/j.cpnec.2022.100143. eCollection 2022 Aug. Compr Psychoneuroendocrinol. 2022. PMID: 35757174 Free PMC article.
-
Comparison of Safety and Efficacy of Methadone vs Morphine for Treatment of Neonatal Abstinence Syndrome: A Randomized Clinical Trial.JAMA Pediatr. 2018 Aug 1;172(8):741-748. doi: 10.1001/jamapediatrics.2018.1307. JAMA Pediatr. 2018. PMID: 29913015 Free PMC article. Clinical Trial.
-
Perinatal oxycodone exposure causes long-term sex-dependent changes in weight trajectory and sensory processing in adult mice.Psychopharmacology (Berl). 2022 Dec;239(12):3859-3873. doi: 10.1007/s00213-022-06257-8. Epub 2022 Oct 21. Psychopharmacology (Berl). 2022. PMID: 36269379
-
Future Newborns with Opioid-Induced Neonatal Abstinence Syndrome (NAS) Could Be Assessed with the Genetic Addiction Risk Severity (GARS) Test and Potentially Treated Using Precision Amino-Acid Enkephalinase Inhibition Therapy (KB220) as a Frontline Modality Instead of Potent Opioids.J Pers Med. 2022 Dec 6;12(12):2015. doi: 10.3390/jpm12122015. J Pers Med. 2022. PMID: 36556236 Free PMC article. Review.
Cited by
-
Advances in animal models of prenatal opioid exposure.Trends Neurosci. 2024 May;47(5):367-382. doi: 10.1016/j.tins.2024.03.005. Epub 2024 Apr 12. Trends Neurosci. 2024. PMID: 38614891 Free PMC article. Review.
References
-
- SAMSHA. Results from the 2016 National Survery on Drug Use and Health: Detailed Tables. 2019.
LinkOut - more resources
Full Text Sources
Miscellaneous

