Myocd regulates airway smooth muscle cell remodeling in response to chronic asthmatic injury
- PMID: 36484734
- PMCID: PMC10107741
- DOI: 10.1002/path.6044
Myocd regulates airway smooth muscle cell remodeling in response to chronic asthmatic injury
Abstract
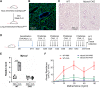
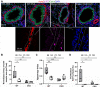
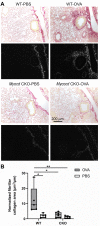
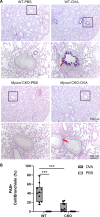
Abnormal growth of airway smooth muscle cells is one of the key features in asthmatic airway remodeling, which is associated with asthma severity. The mechanisms underlying inappropriate airway smooth muscle cell growth in asthma remain largely unknown. Myocd has been reported to act as a key transcriptional coactivator in promoting airway-specific smooth muscle development in fetal lungs. Whether Myocd controls airway smooth muscle remodeling in asthma has not been investigated. Mice with lung mesenchyme-specific deletion of Myocd after lung development were generated, and a chronic asthma model was established by sensitizing and challenging the mice with ovalbumin for a prolonged period. Comparison of the asthmatic pathology between the Myocd knockout mice and the wild-type controls revealed that abrogation of Myocd mitigated airway smooth muscle cell hypertrophy and hyperplasia, accompanied by reduced peri-airway inflammation, decreased fibrillar collagen deposition on airway walls, and attenuation of abnormal mucin production in airway epithelial cells. Our study indicates that Myocd is a key transcriptional coactivator involved in asthma airway remodeling. Inhibition of Myocd in asthmatic airways may be an effective approach to breaking the vicious cycle of asthmatic progression, providing a novel strategy in treating severe and persistent asthma. © 2022 The Authors. The Journal of Pathology published by John Wiley & Sons Ltd on behalf of The Pathological Society of Great Britain and Ireland.
Keywords: Myocd; airway fibrillar collagen; airway inflammation; airway mesenchymal epithelial interaction; airway remodeling; airway smooth muscle cells; asthma.
© 2022 The Authors. The Journal of Pathology published by John Wiley & Sons Ltd on behalf of The Pathological Society of Great Britain and Ireland.
Figures
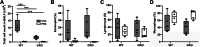

Similar articles
-
The Function of RhoA/ROCK Pathway and MYOCD in Airway Remodeling in Asthma.Int Arch Allergy Immunol. 2025;186(2):103-119. doi: 10.1159/000540963. Epub 2024 Sep 11. Int Arch Allergy Immunol. 2025. PMID: 39260358
-
Contribution of SRF, Elk-1, and myocardin to airway smooth muscle remodeling in heaves, an asthma-like disease of horses.Am J Physiol Lung Cell Mol Physiol. 2015 Jul 1;309(1):L37-45. doi: 10.1152/ajplung.00050.2015. Epub 2015 May 15. Am J Physiol Lung Cell Mol Physiol. 2015. PMID: 25979077
-
Exchange protein directly activated by cAMP (Epac) protects against airway inflammation and airway remodeling in asthmatic mice.Respir Res. 2019 Dec 18;20(1):285. doi: 10.1186/s12931-019-1260-2. Respir Res. 2019. PMID: 31852500 Free PMC article.
-
Rho/ROCK-MYOCD in regulating airway smooth muscle growth and remodeling.Am J Physiol Lung Cell Mol Physiol. 2021 Jul 1;321(1):L1-L5. doi: 10.1152/ajplung.00034.2021. Epub 2021 Apr 28. Am J Physiol Lung Cell Mol Physiol. 2021. PMID: 33909498 Review.
-
Origins of increased airway smooth muscle mass in asthma.BMC Med. 2013 Jun 6;11:145. doi: 10.1186/1741-7015-11-145. BMC Med. 2013. PMID: 23742314 Free PMC article. Review.
Cited by
-
MYOCD and SRF-mediated MLCK transcription prevents polymorphonuclear neutrophils from ferroptosis in sepsis-related acute lung injury.Immunol Res. 2024 Dec;72(6):1299-1312. doi: 10.1007/s12026-024-09529-x. Epub 2024 Aug 28. Immunol Res. 2024. PMID: 39196520
-
Neurodegenerative diseases and neuroinflammation-induced apoptosis.Open Life Sci. 2025 Feb 25;20(1):20221051. doi: 10.1515/biol-2022-1051. eCollection 2025. Open Life Sci. 2025. PMID: 40026360 Free PMC article. Review.
-
Formoterol, the Most Effective Bronchodilator, Has No Anti-Inflammatory nor Metabolic Modulatory Effects in Severe Asthma Induced by Aspergillus fumigatus.ACS Pharmacol Transl Sci. 2025 May 20;8(6):1556-1566. doi: 10.1021/acsptsci.4c00672. eCollection 2025 Jun 13. ACS Pharmacol Transl Sci. 2025. PMID: 40534662
-
Kiss1 receptor knockout exacerbates airway hyperresponsiveness and remodeling in a mouse model of allergic asthma.Respir Res. 2024 Oct 28;25(1):387. doi: 10.1186/s12931-024-03017-4. Respir Res. 2024. PMID: 39468619 Free PMC article.
-
The miR-9-5p/KLF5/IL-1β Axis Regulates Airway Smooth Muscle Cell Proliferation and Apoptosis to Aggravate Airway Remodeling and Inflammation in Asthma.Biochem Genet. 2024 Oct;62(5):3996-4010. doi: 10.1007/s10528-023-10640-1. Epub 2024 Jan 24. Biochem Genet. 2024. PMID: 38267617
References
-
- Payne DN, Rogers AV, Adelroth E, et al. Early thickening of the reticular basement membrane in children with difficult asthma. AmJRespirCrit Care Med 2003; 167: 78–82. - PubMed
-
- Banno A, Reddy AT, Lakshmi SP, et al. Bidirectional interaction of airway epithelial remodeling and inflammation in asthma. Clin Sci (Lond) 2020; 134: 1063–1079. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

