Modulation of the Microglial Nogo-A/NgR Signaling Pathway as a Therapeutic Target for Multiple Sclerosis
- PMID: 36497029
- PMCID: PMC9737582
- DOI: 10.3390/cells11233768
Modulation of the Microglial Nogo-A/NgR Signaling Pathway as a Therapeutic Target for Multiple Sclerosis
Abstract
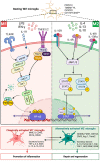
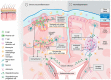
Current therapeutics targeting chronic phases of multiple sclerosis (MS) are considerably limited in reversing the neural damage resulting from repeated inflammation and demyelination insults in the multi-focal lesions. This inflammation is propagated by the activation of microglia, the endogenous immune cell aiding in the central nervous system homeostasis. Activated microglia may transition into polarized phenotypes; namely, the classically activated proinflammatory phenotype (previously categorized as M1) and the alternatively activated anti-inflammatory phenotype (previously, M2). These transitional microglial phenotypes are dynamic states, existing as a continuum. Shifting microglial polarization to an anti-inflammatory status may be a potential therapeutic strategy that can be harnessed to limit neuroinflammation and further neurodegeneration in MS. Our research has observed that the obstruction of signaling by inhibitory myelin proteins such as myelin-associated inhibitory factor, Nogo-A, with its receptor (NgR), can regulate microglial cell function and activity in pre-clinical animal studies. Our review explores the microglial role and polarization in MS pathology. Additionally, the potential therapeutics of targeting Nogo-A/NgR cellular mechanisms on microglia migration, polarization and phagocytosis for neurorepair in MS and other demyelination diseases will be discussed.
Keywords: Nogo receptor; Nogo-A; alternatively activated microglia; classically activated microglia; microglia; microglial polarization; multiple sclerosis; myelin debris.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Cadavid D., Cohen J.A., Freedman M.S., Goldman M.D., Hartung H.P., Havrdova E., Jeffery D., Kapoor R., Miller A., Sellebjerg F., et al. The EDSS-Plus, an improved endpoint for disability progression in secondary progressive multiple sclerosis. Mult. Scler. 2017;23:94–105. doi: 10.1177/1352458516638941. - DOI - PubMed
-
- Pencea V., Bingaman K.D., Wiegand S.J., Luskin M.B. Infusion of Brain-Derived Neurotrophic Factor into the Lateral Ventricle of the Adult Rat Leads to New Neurons in the Parenchyma of the Striatum, Septum, Thalamus, and Hypothalamus. J. Neurosci. 2001;21:6706. doi: 10.1523/JNEUROSCI.21-17-06706.2001. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

