Similarity-Based Virtual Screening to Find Antituberculosis Agents Based on Novel Scaffolds: Design, Syntheses and Pharmacological Assays
- PMID: 36499384
- PMCID: PMC9737236
- DOI: 10.3390/ijms232315057
Similarity-Based Virtual Screening to Find Antituberculosis Agents Based on Novel Scaffolds: Design, Syntheses and Pharmacological Assays
Abstract
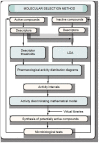
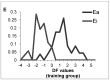
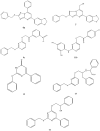
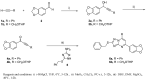
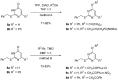
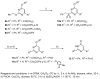
A method to identify molecular scaffolds potentially active against the Mycobacterium tuberculosis complex (MTBC) is developed. A set of structurally heterogeneous agents against MTBC was used to obtain a mathematical model based on topological descriptors. This model was statistically validated through a Leave-n-Out test. It successfully discriminated between active or inactive compounds over 86% in database sets. It was also useful to select new potential antituberculosis compounds in external databases. The selection of new substituted pyrimidines, pyrimidones and triazolo[1,5-a]pyrimidines was particularly interesting because these structures could provide new scaffolds in this field. The seven selected candidates were synthesized and six of them showed activity in vitro.
Keywords: MTBC; antimicrobial drugs; drug design; linear discriminant analysis; pharmacological activity distribution diagrams; topological indices; virtual screening.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
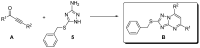
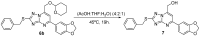
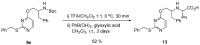
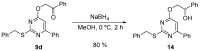
Similar articles
-
Search of chemical scaffolds for novel antituberculosis agents.J Biomol Screen. 2005 Apr;10(3):206-14. doi: 10.1177/1087057104273486. J Biomol Screen. 2005. PMID: 15809316
-
Comparison of Multiple Linear Regressions and Neural Networks based QSAR models for the design of new antitubercular compounds.Eur J Med Chem. 2013;70:831-45. doi: 10.1016/j.ejmech.2013.10.029. Epub 2013 Oct 23. Eur J Med Chem. 2013. PMID: 24246731
-
Global Bayesian models for the prioritization of antitubercular agents.J Chem Inf Model. 2008 Dec;48(12):2362-70. doi: 10.1021/ci800143n. J Chem Inf Model. 2008. PMID: 19053518
-
Applications of Molecular Simulation in the Discovery of Antituberculosis Drugs: A Review.Protein Pept Lett. 2019;26(9):648-663. doi: 10.2174/0929866526666190620145919. Protein Pept Lett. 2019. PMID: 31218945 Review.
-
Current pharmaceutical design of antituberculosis drugs: future perspectives.Curr Pharm Des. 2010;16(24):2656-65. doi: 10.2174/138161210792389289. Curr Pharm Des. 2010. PMID: 20642433 Review.
Cited by
-
Biocompatible 3D-Printed Tendon/Ligament Scaffolds Based on Polylactic Acid/Graphite Nanoplatelet Composites.Nanomaterials (Basel). 2023 Sep 8;13(18):2518. doi: 10.3390/nano13182518. Nanomaterials (Basel). 2023. PMID: 37764548 Free PMC article.
-
Pharmacokinetic Equations Applied to Obtain New Topological Models in the Search of Antibacterial Compounds.Pharmaceuticals (Basel). 2025 Jun 10;18(6):865. doi: 10.3390/ph18060865. Pharmaceuticals (Basel). 2025. PMID: 40573260 Free PMC article.
References
-
- Tuberculosis. [(accessed on 25 June 2022)]. Available online: https://www.who.int/westernpacific/health-topics/tuberculosis.
-
- Dhote A., Kawishwar V. Utility of Concentration Bleach Method and Fluorescent Auramine Rhodamine Staining in Diagnosis of Mycobacterium tuberculosis on Formalin Fixed Tissue. New Horiz. Med. Med. Res. 2022;4:67–70.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

