TiO2 Catalyzed Dihydroxyacetone (DHA) Conversion in Water: Evidence That This Model Reaction Probes Basicity in Addition to Acidity
- PMID: 36500265
- PMCID: PMC9736615
- DOI: 10.3390/molecules27238172
TiO2 Catalyzed Dihydroxyacetone (DHA) Conversion in Water: Evidence That This Model Reaction Probes Basicity in Addition to Acidity
Abstract
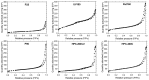
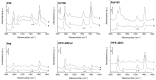
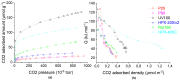
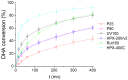
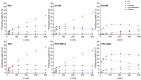
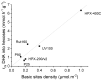
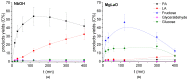
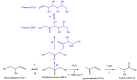
In this paper, evidence is provided that the model reaction of aqueous dihydroxyacetone (DHA) conversion is as sensitive to the TiO2 catalysts' basicity as to their acidity. Two parallel pathways transformed DHA: while the pathway catalyzed by Lewis acid sites gave pyruvaldehyde (PA) and lactic acid (LA), the base-catalyzed route afforded fructose. This is demonstrated on a series of six commercial TiO2 samples and further confirmed by using two reference catalysts: niobic acid (NbOH), an acid catalyst, and a hydrotalcite (MgAlO), a basic catalyst. The original acid-base properties of the six commercial TiO2 with variable structure and texture were investigated first by conventional methods in gas phase (FTIR or microcalorimetry of pyridine, NH3 and CO2 adsorption). A linear relationship between the initial rates of DHA condensation into hexoses and the total basic sites densities is highlighted accounting for the water tolerance of the TiO2 basic sites whatever their strength. Rutile TiO2 samples were the most basic ones. Besides, only the strongest TiO2 Lewis acid sites were shown to be water tolerant and efficient for PA and LA formation.
Keywords: CO2 and NH3 microcalorimetry; FTIR of pyridine adsorption; acidity; basicity; dihydroxyacetone; titanium dioxide.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
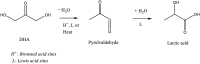






References
-
- Damjanović L., Auroux A. Determination of Acid/Base Properties by Temperature Programmed Desorption (TPD) and Adsorption Calorimetry. In: Chester A.W., Derouane E.G., editors. Zeolite Chemistry and Catalysis. Springer Netherlands; Dordrecht, The Netherlands: 2009. pp. 107–167.
-
- Hunger B., Hoffmann J. Temperature-Programmed Desorption (TPD) of Ammonia from H+-Exchanged Zeolites with Different Structures. J. Therm. Anal. 1988;33:933–940. doi: 10.1007/BF02138612. - DOI
-
- Che M., Vedrine J.C. Characterization of Solid Materials and Heterogeneous Catalysts: From Structure to Surface Reactivity. John Wiley & Sons; Hoboken, NJ, USA: 2012.
-
- Karge H.G. Handbook of Heterogeneous Catalysis. Volume 2. Wiley-VCH; Weinheim, Germany: 2008. Concepts and Analysis of Surface Acidity and Basicity; pp. 1096–1122.
-
- Auroux A. Acidity and Basicity. Volume 6. Springer Berlin Heidelberg; Berlin/Heidelberg, Germany: 2006. Acidity and Basicity: Determination by Adsorption Microcalorimetry; pp. 45–152. Molecular Sieves.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

