Quantum Dot Nanobeads as Multicolor Labels for Simultaneous Multiplex Immunochromatographic Detection of Four Nitrofuran Metabolites in Aquatic Products
- PMID: 36500416
- PMCID: PMC9737793
- DOI: 10.3390/molecules27238324
Quantum Dot Nanobeads as Multicolor Labels for Simultaneous Multiplex Immunochromatographic Detection of Four Nitrofuran Metabolites in Aquatic Products
Abstract
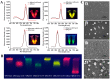
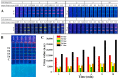
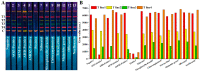
A multicolor immunochromatographic assay platform based on quantum dot nanobeads (QBs) for the rapid and simultaneous detection of nitrofuran metabolites in different aquatic products is documented. These metabolites include 3-amino-2-oxazolidinone (AOZ), 1-aminohydantoin (AHD), semicarbazide (SEM), and 3-amino-5-morpholino-methyl-1,3-oxazolidinone (AMOZ). QBs with emission colors of red, yellow, green, and orange were employed and functionalized with the corresponding antibodies to each analyte to develop a multicolor channel. The visual detection limits (cutoff values) of our method for AOZ, AHD, SEM, and AMOZ reached up to 50 ng/mL, which were 2, 20, 20, and 20 times lower than those of traditional colloidal gold test strips, respectively. The test strip is capable of detection within 10 min in real samples while still achieving good stability and specificity. These results demonstrate that the developed multicolor immunochromatographic assay platform is a promising technique for multiplex, highly sensitive, and on-site detection of nitrofuran metabolites.
Keywords: fluorescent test strip; multiplex immunochromatographic assay; nitrofuran metabolites; quantum dot nanobeads.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
A multiplex immunochromatographic test using gold nanoparticles for the rapid and simultaneous detection of four nitrofuran metabolites in fish samples.Anal Bioanal Chem. 2018 Jan;410(1):223-233. doi: 10.1007/s00216-017-0714-y. Epub 2017 Oct 30. Anal Bioanal Chem. 2018. PMID: 29085985
-
Ratiometric fluorescent immunochromatography for simultaneously detection of two nitrofuran metabolites in seafoods.Food Chem. 2023 Mar 15;404(Pt B):134698. doi: 10.1016/j.foodchem.2022.134698. Epub 2022 Oct 19. Food Chem. 2023. PMID: 36323038
-
Dual-color aggregation-induced emission nanoparticles for simultaneous lateral flow immunoassay of nitrofuran metabolites in aquatic products.Food Chem. 2023 Feb 15;402:134235. doi: 10.1016/j.foodchem.2022.134235. Epub 2022 Sep 14. Food Chem. 2023. PMID: 36155286
-
Simultaneous detection of four nitrofuran metabolites in honey by using a visualized microarray screen assay.Food Chem. 2017 Apr 15;221:1813-1821. doi: 10.1016/j.foodchem.2016.10.099. Epub 2016 Oct 24. Food Chem. 2017. PMID: 27979167
-
Immunosensor of Nitrofuran Antibiotics and Their Metabolites in Animal-Derived Foods: A Review.Front Chem. 2022 Jun 1;10:813666. doi: 10.3389/fchem.2022.813666. eCollection 2022. Front Chem. 2022. PMID: 35721001 Free PMC article. Review.
Cited by
-
Multi-chromatic and multi-component lateral flow immunoassay for simultaneous detection of CP4 EPSPS, Bt-Cry1Ab, Bt-Cry1Ac, and PAT/bar proteins in genetically modified crops.Mikrochim Acta. 2024 Dec 16;192(1):16. doi: 10.1007/s00604-024-06853-9. Mikrochim Acta. 2024. PMID: 39680231
-
A comprehensive review on the pretreatment and detection methods of nitrofurans and their metabolites in animal-derived food and environmental samples.Food Chem X. 2024 Oct 28;24:101928. doi: 10.1016/j.fochx.2024.101928. eCollection 2024 Dec 30. Food Chem X. 2024. PMID: 39539437 Free PMC article. Review.
References
-
- Wu W., Yang S., Liu J., Mi J., Dou L., Pan Y., Mari G.M., Wang Z. Progress in immunoassays for nitrofurans detection. Food Agr. Immunol. 2020;31:907–926. doi: 10.1080/09540105.2020.1786672. - DOI
-
- Luo X., Yu Y., Kong X., Wang X., Ji Z., Sun Z., You J. Rapid microwave assisted derivatization of nitrofuran metabolites for analysis in shrimp by high performance liquid chromatography-fluorescence detector. Microchem. J. 2019;150:104189. doi: 10.1016/j.microc.2019.104189. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

