Promising drugs and treatment options for pediatric and adolescent patients with Hodgkin lymphoma
- PMID: 36506094
- PMCID: PMC9729954
- DOI: 10.3389/fcell.2022.965803
Promising drugs and treatment options for pediatric and adolescent patients with Hodgkin lymphoma
Abstract
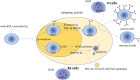
Currently-available therapies for newly-diagnosed pediatric and adolescent patients with Hodgkin lymphoma result in >95% survival at 5 years. Long-term survivors may suffer from long-term treatment-related side effects, however, so the past 20 years have seen clinical trials for children and adolescents with HL gradually abandon the regimens used in adults in an effort to improve this situation. Narrower-field radiotherapy can reduce long-term toxicity while maintaining good tumor control. Various risk-adapted chemo-radiotherapy strategies have been used. Early assessment of tumor response with interim positron emission tomography and/or measuring metabolic tumor volume has been used both to limit RT in patients with favorable characteristics and to adopt more aggressive therapies in patients with a poor response. Most classical Hodgkin's lymphoma relapses occur within 3 years of initial treatment, while relapses occurring 5 years or more after diagnosis are rare. As the outcome for patients with relapsed/refractory classical Hodgkin lymphoma remains unsatisfactory, new drugs have been proposed for its prevention or treatment. This review summarizes the important advances made in recent years in the management of pediatric and adolescent with classical Hodgkin lymphoma, and the novel targeted treatments for relapsed and refractory classical Hodgkin lymphoma.
Keywords: Epstein-Barr virus; Hodgkin lymphoma; adolescent; chemotherapy; radiation; tumor target.
Copyright © 2022 De Re, Repetto, Mussolin, Brisotto, Elia, Lopci, d’Amore, Burnelli and Mascarin.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Aide N., Hicks R. J., Le Tourneau C., Lheureux S., Fanti S., Lopci E. (2019). FDG PET/CT for assessing tumour response to immunotherapy : Report on the EANM symposium on immune modulation and recent review of the literature. Eur. J. Nucl. Med. Mol. Imaging 46, 238–250. 10.1007/s00259-018-4171-4 - DOI - PMC - PubMed
-
- Airtum Working Group, Ccm (2013). Italian cancer figures, report 2012: Cancer in children and adolescents. Epidemiol. Prev. 37, 1–225. - PubMed
-
- Baetz T., Belch A., Couban S., Imrie K., Yau J., Myers R., et al. (2003). Gemcitabine, dexamethasone and cisplatin is an active and non-toxic chemotherapy regimen in relapsed or refractory Hodgkin’s disease: A phase II study by the national cancer institute of Canada clinical trials group. Ann. Oncol. 14, 1762–1767. 10.1093/annonc/mdg496 - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

