An intravenous anesthetic drug-propofol, influences the biological characteristics of malignant tumors and reshapes the tumor microenvironment: A narrative literature review
- PMID: 36506511
- PMCID: PMC9732110
- DOI: 10.3389/fphar.2022.1057571
An intravenous anesthetic drug-propofol, influences the biological characteristics of malignant tumors and reshapes the tumor microenvironment: A narrative literature review
Abstract
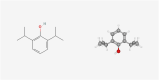
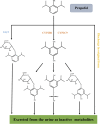
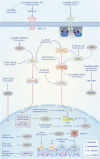
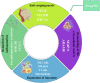
Malignant tumors are the second leading cause of death worldwide. This is a public health concern that negatively impacts human health and poses a threat to the safety of life. Although there are several treatment approaches for malignant tumors, surgical resection remains the primary and direct treatment for malignant solid tumors. Anesthesia is an integral part of the operation process. Different anesthesia techniques and drugs have different effects on the operation and the postoperative prognosis. Propofol is an intravenous anesthetic that is commonly used in surgery. A substantial number of studies have shown that propofol participates in the pathophysiological process related to malignant tumors and affects the occurrence and development of malignant tumors, including anti-tumor effect, pro-tumor effect, and regulation of drug resistance. Propofol can also reshape the tumor microenvironment, including anti-angiogenesis, regulation of immunity, reduction of inflammation and remodeling of the extracellular matrix. Furthermore, most clinical studies have also indicated that propofol may contribute to a better postoperative outcome in some malignant tumor surgeries. Therefore, the author reviewed the chemical properties, pharmacokinetics, clinical application and limitations, mechanism of influencing the biological characteristics of malignant tumors and reshaping the tumor microenvironment, studies of propofol in animal tumor models and its relationship with postoperative prognosis of propofol in combination with the relevant literature in recent years, to lay a foundation for further study on the correlation between propofol and malignant tumor and provide theoretical guidance for the selection of anesthetics in malignant tumor surgery.
Keywords: anesthetic drug; biological characteristics; chemical properties; malignant tumor; pharmacokinetic; postoperative prognosis; propofol; tumor microenvironment.
Copyright © 2022 Zhou, Shao, Li, Zhang, Ding, Zhuang and Sun.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Effects of Propofol Versus Sevoflurane on Postoperative Breast Cancer Prognosis: A Narrative Review.Front Oncol. 2022 Jan 20;11:793093. doi: 10.3389/fonc.2021.793093. eCollection 2021. Front Oncol. 2022. PMID: 35127500 Free PMC article. Review.
-
Effectiveness of ondansetron as an adjunct to lidocaine intravenous regional anesthesia on tourniquet pain and postoperative pain in patients undergoing elective hand surgery: a systematic review protocol.JBI Database System Rev Implement Rep. 2015 Jan;13(1):27-38. doi: 10.11124/jbisrir-2015-1768. JBI Database System Rev Implement Rep. 2015. PMID: 26447005
-
Propofol anesthesia.Vet Clin North Am Small Anim Pract. 1999 May;29(3):747-78. doi: 10.1016/s0195-5616(99)50059-4. Vet Clin North Am Small Anim Pract. 1999. PMID: 10332821 Review.
-
The benefits of propofol on cancer treatment: Decipher its modulation code to immunocytes.Front Pharmacol. 2022 Nov 4;13:919636. doi: 10.3389/fphar.2022.919636. eCollection 2022. Front Pharmacol. 2022. PMID: 36408275 Free PMC article. Review.
-
Propofol and Sevoflurane Differentially Impact MicroRNAs in Circulating Extracellular Vesicles during Colorectal Cancer Resection: A Pilot Study.Anesthesiology. 2020 Jan;132(1):107-120. doi: 10.1097/ALN.0000000000002986. Anesthesiology. 2020. PMID: 31596735
Cited by
-
Predictive value of long non-coding RNA DDX11-AS1 in inflammatory bowel disease and its effect on intestinal mucosal cell function.Cent Eur J Immunol. 2025;50(1):87-97. doi: 10.5114/ceji.2025.149579. Epub 2025 Apr 16. Cent Eur J Immunol. 2025. PMID: 40620651 Free PMC article.
-
Impact of propofol on gastrointestinal cancer outcomes: A review of cellular behavior, growth, and metastasis.World J Clin Oncol. 2025 Jul 24;16(7):104727. doi: 10.5306/wjco.v16.i7.104727. World J Clin Oncol. 2025. PMID: 40741181 Free PMC article. Review.
-
The synergistic mechanisms of propofol with cisplatin or doxorubicin in human ovarian cancer cells.J Ovarian Res. 2024 Sep 14;17(1):187. doi: 10.1186/s13048-024-01509-x. J Ovarian Res. 2024. PMID: 39272193 Free PMC article.
-
Influence of the Anesthetic Technique on Circulating Extracellular Vesicles in Bladder Cancer Patients Undergoing Radical Cystectomy: A Prospective, Randomized Trial.Cells. 2023 Oct 23;12(20):2503. doi: 10.3390/cells12202503. Cells. 2023. PMID: 37887347 Free PMC article. Clinical Trial.
-
The Inhibitory Effects of Propofol on Colorectal Cancer Progression through the NF-κB/HIF-1α Signaling Pathway.Anticancer Agents Med Chem. 2024;24(11):878-888. doi: 10.2174/0118715206283884240326170501. Anticancer Agents Med Chem. 2024. PMID: 38571352
References
Publication types
LinkOut - more resources
Full Text Sources

