Doxycycline attenuates l-DOPA-induced dyskinesia through an anti-inflammatory effect in a hemiparkinsonian mouse model
- PMID: 36506543
- PMCID: PMC9728610
- DOI: 10.3389/fphar.2022.1045465
Doxycycline attenuates l-DOPA-induced dyskinesia through an anti-inflammatory effect in a hemiparkinsonian mouse model
Abstract
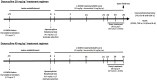
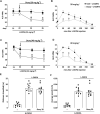
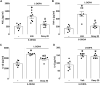
The pharmacological manipulation of neuroinflammation appears to be a promising strategy to alleviate l-DOPA-induced dyskinesia (LID) in Parkinson's disease (PD). Doxycycline (Doxy), a semisynthetic brain-penetrant tetracycline antibiotic having interesting anti-inflammatory properties, we addressed the possibility that this compound could resolve LID in l-DOPA-treated C57BL/6 mice presenting either moderate or intermediate lesions of the mesostriatal dopaminergic pathway generated by intrastriatal injections of 6-OHDA. Doxy, when given subcutaneously before l-DOPA at doses of 20 mg kg-1 and 40 mg kg-1, led to significant LID reduction in mice with moderate and intermediate dopaminergic lesions, respectively. Importantly, Doxy did not reduce locomotor activity improved by l-DOPA. To address the molecular mechanism of Doxy, we sacrificed mice with mild lesions 1) to perform the immunodetection of tyrosine hydroxylase (TH) and Fos-B and 2) to evaluate a panel of inflammation markers in the striatum, such as cyclooxygenase-2 and its downstream product Prostaglandin E2 along with the cytokines TNF-α, IL-1β and IL-6. TH-immunodetection revealed that vehicle and Doxy-treated mice had similar striatal lesions, excluding that LID improvement by Doxy could result from neurorestorative effects. Importantly, LID inhibition by Doxy was associated with decreased Fos-B and COX-2 expression and reduced levels of PGE2, TNF-α, and IL-1β in the dorsolateral striatum of dyskinetic mice. We conclude 1) that Doxy has the potential to prevent LID regardless of the intensity of dopaminergic lesioning and 2) that the anti-inflammatory effects of Doxy probably account for LID attenuation. Overall, the present results further indicate that Doxy might represent an attractive and alternative treatment for LID in PD.
Keywords: COX-2; IL-1β; PGE2; Parkinson’s disease; TNF-α; cytokines; neuroinflammation; tetracyclines.
Copyright © 2022 dos Santos Pereira, Nascimento, Bortolanza, Michel, Raisman-Vozari and Del Bel.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Differential induction of dyskinesia and neuroinflammation by pulsatile versus continuous l-DOPA delivery in the 6-OHDA model of Parkinson's disease.Exp Neurol. 2016 Dec;286:83-92. doi: 10.1016/j.expneurol.2016.09.013. Epub 2016 Sep 30. Exp Neurol. 2016. PMID: 27697481
-
Treadmill Exercise Attenuates L-DOPA-Induced Dyskinesia and Increases Striatal Levels of Glial Cell-Derived Neurotrophic Factor (GDNF) in Hemiparkinsonian Mice.Mol Neurobiol. 2019 Apr;56(4):2944-2951. doi: 10.1007/s12035-018-1278-3. Epub 2018 Aug 2. Mol Neurobiol. 2019. PMID: 30073506
-
Angiotensin type 1 receptor blockage reduces l-dopa-induced dyskinesia in the 6-OHDA model of Parkinson's disease. Involvement of vascular endothelial growth factor and interleukin-1β.Exp Neurol. 2014 Nov;261:720-32. doi: 10.1016/j.expneurol.2014.08.019. Epub 2014 Aug 23. Exp Neurol. 2014. PMID: 25160895
-
Neuroinflammation in L-DOPA-induced dyskinesia: beyond the immune function.J Neural Transm (Vienna). 2018 Aug;125(8):1287-1297. doi: 10.1007/s00702-018-1874-4. Epub 2018 Mar 14. J Neural Transm (Vienna). 2018. PMID: 29541852 Review.
-
Animal models of L-DOPA-induced dyskinesia: the 6-OHDA-lesioned rat and mouse.J Neural Transm (Vienna). 2018 Aug;125(8):1137-1144. doi: 10.1007/s00702-017-1825-5. Epub 2017 Dec 14. J Neural Transm (Vienna). 2018. PMID: 29242978 Review.
Cited by
-
Doxycycline: An essential tool for Alzheimer's disease.Biomed Pharmacother. 2025 Jul;188:118159. doi: 10.1016/j.biopha.2025.118159. Epub 2025 May 13. Biomed Pharmacother. 2025. PMID: 40367557 Free PMC article. Review.
-
Exploring the causal relationship between B lymphocytes and Parkinson's disease: a bidirectional, two-sample Mendelian randomization study.Sci Rep. 2024 Feb 2;14(1):2783. doi: 10.1038/s41598-024-53287-7. Sci Rep. 2024. PMID: 38307922 Free PMC article.
-
Cannabigerol Mitigates Haloperidol-Induced Vacuous Chewing Movements in Mice.Neurotox Res. 2024 Dec 19;43(1):2. doi: 10.1007/s12640-024-00724-0. Neurotox Res. 2024. PMID: 39699828
-
Alpha Synuclein: Neurodegeneration and Inflammation.Int J Mol Sci. 2023 Mar 21;24(6):5914. doi: 10.3390/ijms24065914. Int J Mol Sci. 2023. PMID: 36982988 Free PMC article. Review.
-
Histopathologic evaluation of skin wound healing due to local application of transdermal chitosan patch in combination with doxycycline, zinc nanoparticles, and selenium nanoparticles in mice.BMC Vet Res. 2025 Mar 31;21(1):221. doi: 10.1186/s12917-025-04672-5. BMC Vet Res. 2025. PMID: 40159480 Free PMC article.
References
-
- Andersson H., Alestig K. (1976). The penetration of doxycycline into CSF. Scand. J. Infect. Dis. Suppl. Suppl, 17–19. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

