Thymoquinone protects the testes of hypothyroid rats by suppressing pro-inflammatory cytokines and oxidative stress and promoting SIRT1 testicular expression
- PMID: 36506574
- PMCID: PMC9731332
- DOI: 10.3389/fphar.2022.1040857
Thymoquinone protects the testes of hypothyroid rats by suppressing pro-inflammatory cytokines and oxidative stress and promoting SIRT1 testicular expression
Abstract
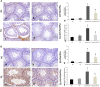
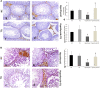
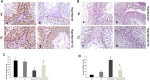
Background: Hypothyroidism has been linked to many testicular structural and dysfunctional changes in males. Thymoquinone (TQ) has shown a potent testicular protective effect through its antioxidant, anti-inflammatory, antiapoptotic, fertility-enhancing, and endocrine modulatory activities. Objectives: This study aimed to investigate the efficacy of TQ in preserving the testicular structure of a model of experimentally induced hypothyroidism in rats and identify the mechanism behind this effect. Materials and methods: Propylthiouracil (PTU) was used to induce hypothyroidism in adult male Wistar rats, who were then treated with TQ (50 mg/kg/body weight) for 4 weeks and compared to the untreated rats. Thyroid hormonal profile, oxidants/antioxidants profile, and serum testosterone levels were assessed. Gene expression and immune expression of SIRT1 and pro-inflammatory cytokines TNF-α and NF-κB were also assessed in the testicular tissue. Results: TQ administration successfully improved PTU-induced disturbance in the thyroid hormonal profile (T3, T4, and TSH), serum testosterone level, and pancreatic antioxidants compared to the untreated hypothyroid group. TQ significantly downregulated (p = 0.001, p ˂ 0.001) TNF-α and NF-κB transcription, while it significantly upregulated (p = 0.01) SIRT1 transcription in the testes of hypothyroid rats. TQ markedly relieved the histopathological testicular changes induced by PTU and significantly increased (p = 0.002, p = 0.01) the sectional area of seminiferous tubules and germinal epithelial height, respectively. TUNEL-positive apoptotic germinal cells were significantly decreased (p ˂ 0.001), while PCNA-positive proliferating germinal cells and androgen receptor expression were significantly increased (p ˂ 0.001) in the testes of TQ-treated hypothyroid rats. Conclusion: Thymoquinone could limit the hypothyroidism-induced structural changes in the testis, mostly through the upregulation of SIRT1 expression, which seems to mediate its promising antioxidant, anti-inflammatory and antiapoptotic effects that were evident in this study. Therefore, TQ is recommended as an adjuvant safe supplement in managing hypothyroidism, especially in males.
Keywords: NF-κB; PCNA; SIRT1; TNF-α; hypothyroidism; rat; testis; thymoquinone.
Copyright © 2022 Algaidi, Faddladdeen, Alrefaei, Qahl, Albadawi, ALmohaimeed and Ayuob.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
-
- Aithal M., Shaik H., Das K. K., Saheb H. (2016). Effect of Nigella sativa seed and thymoquinone on reproductive parameters in sterptozotocine induced diabetic and normal male albino rats. Int. J. Intg Med. Sci. 3, 248–252. 10.16965/ijims.2016.109 - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous

