Adverse event profiles of adjuvant treatment with opicapone in Parkinson's disease: A systematic review and meta-analysis
- PMID: 36506576
- PMCID: PMC9729693
- DOI: 10.3389/fphar.2022.1042992
Adverse event profiles of adjuvant treatment with opicapone in Parkinson's disease: A systematic review and meta-analysis
Abstract
Background: Opicapone, a novel third-generation catechol-O-methyltransferase inhibitor, has demonstrated efficacy in Parkinson's Disease (PD) patients with end-of-dose motor fluctuations. Objective: This study aimed to compare the short-term (<6 months) and long-term (≥6 months) tolerability of opicapone adjuvant treatment in PD patients. Method: Electronic databases including PubMed, Embase, Web of Science and Cochrane library were searched for randomized controlled trials (RCTs) and observational studies. The end points included any treatment-related adverse events (TEAEs), serious TEAEs (SAEs) and treatment discontinuation. A random-effects model was used to generate overall incidences of TEAE. Results: Three RCTs, three RCT extension studies and three open-label studies involving 2177 PD patients were evaluated. In the short-term studies, there were reports of TEAEs with an incidence of ≥5% in individuals treated with opicapone 50 mg, including dyskinesia (14.1%), elevated blood creatine phosphokinase levels (8.0%) and urinary tract infection (6.0%). Any TEAEs, SAEs and treatment discontinuation all occurred at rates of 62.9%, 4.8% and 9.3%, respectively. TEAEs with opicapone 50 mg that were reported by more than 5% of patients in long-term studies included dyskinesia (16.1%), dry mouth (12.1%), medication effect decreased (12.1%), PD exacerbated (7.8%), blood creatine phosphokinase level raised (7.4%), nausea (6.1%) and insomnia (5.1%). The incidence of any TEAEs, SAEs and treatment discontinuation were, correspondingly, 73.2%, 8.7% and 8.4%. Conclusion: These studies demonstrated that opicapone was generally well-tolerated and had a low risk of adverse events, suggesting that it could be a valuable therapeutic choice for people with PD.
Keywords: Parkinson’s disease; adjunctive treatment; adverse events; meta-analysis; opicapone.
Copyright © 2022 Xie, Qi, Wang, He, Wang, Zhang, Yu, Deng, Liang and Lü.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
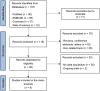
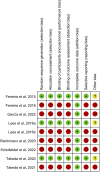


References
-
- Bonifácio M. J., Sutcliffe J. S., Torrão L., Wright L. C., Soares-da-Silva P. (2014). Brain and peripheral pharmacokinetics of levodopa in the cynomolgus monkey following administration of opicapone, a third generation nitrocatechol COMT inhibitor. Neuropharmacology 77, 334–341. 10.1016/j.neuropharm.2013.10.014 - DOI - PubMed
-
- Chong D. J., Suchowersky O., Szumlanski C., Weinshilboum R. M., Brant R., Campbell N. R. (2000). The relationship between COMT genotype and the clinical effectiveness of tolcapone, a COMT inhibitor, in patients with Parkinson's disease. Clin. Neuropharmacol. 23, 143–148. 10.1097/00002826-200005000-00003 - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources

