Small-molecule PROTAC mediates targeted protein degradation to treat STAT3-dependent epithelial cancer
- PMID: 36509291
- PMCID: PMC9746828
- DOI: 10.1172/jci.insight.160606
Small-molecule PROTAC mediates targeted protein degradation to treat STAT3-dependent epithelial cancer
Abstract
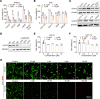
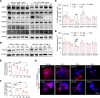
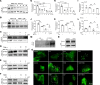
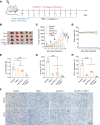
The aberrant activation of STAT3 is associated with the etiology and progression in a variety of malignant epithelial-derived tumors, including head and neck squamous cell carcinoma (HNSCC) and colorectal cancer (CRC). Due to the lack of an enzymatic catalytic site or a ligand-binding pocket, there are no small-molecule inhibitors directly targeting STAT3 that have been approved for clinical translation. Emerging proteolysis targeting chimeric (PROTAC) technology-based approach represents a potential strategy to overcome the limitations of conventional inhibitors and inhibit activation of STAT3 and downstream genes. In this study, the heterobifunctional small-molecule-based PROTACs are successfully prepared from toosendanin (TSN), with 1 portion binding to STAT3 and the other portion binding to an E3 ubiquitin ligase. The optimized lead PROTAC (TSM-1) exhibits superior selectivity, potency, and robust antitumor effects in STAT3-dependent HNSCC and CRC - especially in clinically relevant patient-derived xenografts (PDX) and patient-derived organoids (PDO). The following mechanistic investigation identifies the reduced expression of critical downstream STAT3 effectors, through which TSM-1 promotes cell cycle arrest and apoptosis in tumor cells. These findings provide the first demonstration to our knowledge of a successful PROTAC-targeting strategy in STAT3-dependent epithelial cancer.
Keywords: Colorectal cancer; Drug therapy; Head and neck cancer; Oncology; Therapeutics.
Figures





References
-
- Kusaba T, et al. Activation of STAT3 is a marker of poor prognosis in human colorectal cancer. Oncol Rep. 2006;15(6):1445–1451. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

