Upper gut heat shock proteins HSP70 and GRP78 promote insulin resistance, hyperglycemia, and non-alcoholic steatohepatitis
- PMID: 36513656
- PMCID: PMC9748124
- DOI: 10.1038/s41467-022-35310-5
Upper gut heat shock proteins HSP70 and GRP78 promote insulin resistance, hyperglycemia, and non-alcoholic steatohepatitis
Abstract
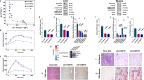
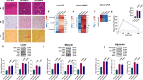
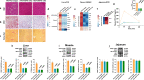
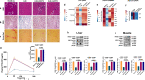
A high-fat diet increases the risk of insulin resistance, type-2 diabetes, and non-alcoholic steato-hepatitis. Here we identified two heat-shock proteins, Heat-Shock-Protein70 and Glucose-Regulated Protein78, which are increased in the jejunum of rats on a high-fat diet. We demonstrated a causal link between these proteins and hepatic and whole-body insulin-resistance, as well as the metabolic response to bariatric/metabolic surgery. Long-term continuous infusion of Heat-Shock-Protein70 and Glucose-Regulated Protein78 caused insulin-resistance, hyperglycemia, and non-alcoholic steato-hepatitis in rats on a chow diet, while in rats on a high-fat diet continuous infusion of monoclonal antibodies reversed these phenotypes, mimicking metabolic surgery. Infusion of these proteins or their antibodies was also associated with shifts in fecal microbiota composition. Serum levels of Heat-Shock-Protein70 and Glucose-Regulated Protein78were elevated in patients with non-alcoholic steato-hepatitis, but decreased following metabolic surgery. Understanding the intestinal regulation of metabolism may provide options to reverse metabolic diseases.
© 2022. The Author(s).
Conflict of interest statement
G.M. reports consulting fees from Novo Nordisk, Fractyl Inc, Recor Inc. She is also Scientific Advisor of Keyron Ltd, Metadeq Inc, GHP Scientific Ltd, Jemyll Ltd. F.R. reports receiving research grants from Ethicon and Medtronic; receiving consulting fees from Novo Nordisk, Ethicon, and Medtronic; and serving on scientific advisory boards for GI Dynamics and Keyron. C.W.l.R. reports grants from the Irish Research Council, Science Foundation Ireland, Anabio, and the Health Research Board. He serves on advisory boards of Novo Nordisk, Herbalife, GI Dynamics, Eli Lilly, Johnson & Johnson, Sanofi Aventis, AstraZeneca, Janssen, Bristol-Myers Squibb, Glia, and Boehringer Ingelheim. C.W.l.R. is a member of the Irish Society for Nutrition and Metabolism outside the area of work commented on here. He is the chief medical officer and director of the Medical Device Division of Keyron since January 2011. Both of these are unremunerated positions. C.W.l.R. was a previous investor in Keyron, which develops endoscopically implantable medical devices intended to mimic the surgical procedures of sleeve gastrectomy and gastric bypass. The product has only been tested in rodents and none of Keyron’s products are currently licensed. They do not have any contracts with other companies to put their products into clinical practice. No patients have been included in any of Keyron’s studies and they are not listed on the stock market. He continues to provide scientific advice to Keyron for no remuneration. The remaining authors declare no competing interests.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

