Single nuclei profiling identifies cell specific markers of skeletal muscle aging, frailty, and senescence
- PMID: 36516485
- PMCID: PMC9792217
- DOI: 10.18632/aging.204435
Single nuclei profiling identifies cell specific markers of skeletal muscle aging, frailty, and senescence
Abstract
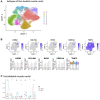
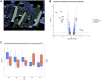
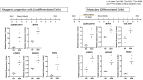
Aging is accompanied by a loss of muscle mass and function, termed sarcopenia, which causes numerous morbidities and economic burdens in human populations. Mechanisms implicated in age-related sarcopenia or frailty include inflammation, muscle stem cell depletion, mitochondrial dysfunction, and loss of motor neurons, but whether there are key drivers of sarcopenia are not yet known. To gain deeper insights into age-related muscle loss, we performed transcriptome profiling on lower limb muscle biopsies from 72 young, elderly, and frail human subjects using bulk RNA-seq (N = 72) and single-nuclei RNA-seq (N = 17). This combined approach revealed changes in gene expression that occur with age and frailty in multiple cell types comprising mature skeletal muscle. Notably, we found increased expression of the genes MYH8 and PDK4, and decreased expression of the gene IGFN1, in aged muscle. We validated several key genes changes in fixed human muscle tissue using digital spatial profiling. We also identified a small population of nuclei that express CDKN1A, present only in aged samples, consistent with p21cip1-driven senescence in this subpopulation. Overall, our findings identify unique cellular subpopulations in aged and sarcopenic skeletal muscle, which will facilitate the development of new therapeutic strategies to combat age-related frailty.
Keywords: aging; muscle; sarcopenia; senescence; transcriptomics.
Conflict of interest statement
Figures




References
-
- Koster A, Ding J, Stenholm S, Caserotti P, Houston DK, Nicklas BJ, You T, Lee JS, Visser M, Newman AB, Schwartz AV, Cauley JA, Tylavsky FA, et al. , and Health ABC study. Does the amount of fat mass predict age-related loss of lean mass, muscle strength, and muscle quality in older adults? J Gerontol A Biol Sci Med Sci. 2011; 66:888–95. 10.1093/gerona/glr070 - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

