The role of bacterial vaccines in the fight against antimicrobial resistance: an analysis of the preclinical and clinical development pipeline
- PMID: 36528040
- PMCID: PMC9892012
- DOI: 10.1016/S2666-5247(22)00303-2
The role of bacterial vaccines in the fight against antimicrobial resistance: an analysis of the preclinical and clinical development pipeline
Abstract
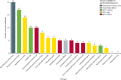
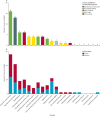
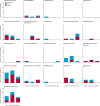
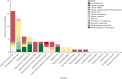
Vaccines can be highly effective tools in combating antimicrobial resistance as they reduce infections caused by antibiotic-resistant bacteria and antibiotic consumption associated with disease. This Review looks at vaccine candidates that are in development against pathogens on the 2017 WHO bacterial priority pathogen list, in addition to Clostridioides difficile and Mycobacterium tuberculosis. There were 94 active preclinical vaccine candidates and 61 active development vaccine candidates. We classified the included pathogens into the following four groups: Group A consists of pathogens for which vaccines already exist-ie, Salmonella enterica serotype Typhi, Streptococcus pneumoniae, Haemophilus influenzae type b, and M tuberculosis. Group B consists of pathogens with vaccines in advanced clinical development-ie, extra-intestinal pathogenic Escherichia coli, Salmonella enterica serotype Paratyphi A, Neisseria gonorrhoeae, and C difficile. Group C consists of pathogens with vaccines in early phases of clinical development-ie, enterotoxigenic E coli, Klebsiella pneumoniae, non-typhoidal Salmonella, Shigella spp, and Campylobacter spp. Finally, group D includes pathogens with either no candidates in clinical development or low development feasibility-ie, Pseudomonas aeruginosa, Acinetobacter baumannii, Staphylococcus aureus, Helicobacter pylori, Enterococcus faecium, and Enterobacter spp. Vaccines are already important tools in reducing antimicrobial resistance and future development will provide further opportunities to optimise the use of vaccines against resistance.
Copyright © 2023 World Health Organization; licensee Elsevier. This is an Open Access article published under the CC BY 3.0 IGO license which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. In any use of this article, there should be no suggestion that WHO endorses any specific organisation, products or services. The use of the WHO logo is not permitted. This notice should be preserved along with the article's original URL.
Conflict of interest statement
Declaration of interests We declare no competing interests.
Figures

Similar articles
-
Synthetic Glycans to Improve Current Glycoconjugate Vaccines and Fight Antimicrobial Resistance.Chem Rev. 2022 Oct 26;122(20):15672-15716. doi: 10.1021/acs.chemrev.2c00021. Epub 2022 May 24. Chem Rev. 2022. PMID: 35608633 Free PMC article. Review.
-
Bacterial distribution and drug resistance in blood samples of children in Jiangxi Region, 2017-2021.Front Cell Infect Microbiol. 2023 Jun 22;13:1163312. doi: 10.3389/fcimb.2023.1163312. eCollection 2023. Front Cell Infect Microbiol. 2023. PMID: 37424793 Free PMC article.
-
Vaccines for viral and bacterial pathogens causing acute gastroenteritis: Part II: Vaccines for Shigella, Salmonella, enterotoxigenic E. coli (ETEC) enterohemorragic E. coli (EHEC) and Campylobacter jejuni.Hum Vaccin Immunother. 2015;11(3):601-19. doi: 10.1080/21645515.2015.1011578. Hum Vaccin Immunother. 2015. PMID: 25715096 Free PMC article. Review.
-
Antibiotic Resistance Trends in ESKAPE Pathogens Isolated at a Health Practice and Research Hospital: A Five-Year Retrospective Study.J Infect Dev Ctries. 2024 Dec 30;18(12):1899-1908. doi: 10.3855/jidc.19592. J Infect Dev Ctries. 2024. PMID: 39832249
-
Discovery, research, and development of new antibiotics: the WHO priority list of antibiotic-resistant bacteria and tuberculosis.Lancet Infect Dis. 2018 Mar;18(3):318-327. doi: 10.1016/S1473-3099(17)30753-3. Epub 2017 Dec 21. Lancet Infect Dis. 2018. PMID: 29276051
Cited by
-
Healthcare as a driver, reservoir and amplifier of antimicrobial resistance: opportunities for interventions.Nat Rev Microbiol. 2024 Oct;22(10):636-649. doi: 10.1038/s41579-024-01076-4. Epub 2024 Jul 24. Nat Rev Microbiol. 2024. PMID: 39048837 Review.
-
Trivalent outer membrane vesicles-based combination vaccine candidate induces protective immunity against Campylobacter and invasive non-typhoidal Salmonella in adult mice.Med Microbiol Immunol. 2024 Oct 15;213(1):21. doi: 10.1007/s00430-024-00805-z. Med Microbiol Immunol. 2024. PMID: 39407046
-
Immune interface interference vaccines: An evolution-informed approach to anti-bacterial vaccine design.Microb Biotechnol. 2024 Mar;17(3):e14446. doi: 10.1111/1751-7915.14446. Microb Biotechnol. 2024. PMID: 38536702 Free PMC article.
-
Challenges and opportunities in mRNA vaccine development against bacteria.Nat Microbiol. 2025 Aug;10(8):1816-1828. doi: 10.1038/s41564-025-02070-z. Epub 2025 Jul 29. Nat Microbiol. 2025. PMID: 40730911 Review.
-
Therapeutic Strategies to Combat Increasing Rates of Multidrug Resistant Pathogens.Pharm Res. 2024 Aug;41(8):1557-1571. doi: 10.1007/s11095-024-03756-5. Epub 2024 Aug 6. Pharm Res. 2024. PMID: 39107513 Review.
References
-
- Europe RAND . Wellcome Trust; London: 2021. How drug-resistant infections are undermining modern medicine—and why more research is needed now.
-
- Gooch J, Rodriguez A, Serazin E, Schwarm M, Steene A, Cheng YY. Vaccines to tackle drug resistant infections: an evaluation of R&D opportunities. Oct 6, 2021. https://www.bcg.com/vaccines-to-tackle-drug-resistant-infections
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

