Oroxin B alleviates osteoarthritis through anti-inflammation and inhibition of PI3K/AKT/mTOR signaling pathway and enhancement of autophagy
- PMID: 36531454
- PMCID: PMC9751055
- DOI: 10.3389/fendo.2022.1060721
Oroxin B alleviates osteoarthritis through anti-inflammation and inhibition of PI3K/AKT/mTOR signaling pathway and enhancement of autophagy
Abstract
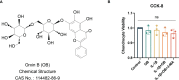
Background: Osteoarthritis (OA) is a common aging-related degenerative joint disease with chronic inflammation as its possible pathogenesis. Oroxin B (OB), a flavonoid isolated from traditional Chinese herbal medicine, possesses anti-inflammation properties which may be involved in regulating the pathogenesis of OA, but its mechanism has not been elucidated. Our study was the first to explore the potential chondroprotective effect and elucidate the underlying mechanism of OB in OA.
Methods: In vitro, primary mice chondrocytes were stimulated with IL-1β along with or without the administration of OB or autophagy inhibitor 3-methyladenine (3-MA). Cell viability assay was measured with a cell counting kit-8 (CCK-8). The phenotypes of anabolic-related (Aggrecan and Collagen II), catabolic-related (MMP3, MMP13, and ADAMTS5), inflammation-related (iNOS, COX-2, TNF-α, IL-6, and IL-1β), and markers of related signaling pathways in chondrocytes with different treatment were detected through western blot, RT-qPCR, and immunofluorescent staining. In vivo, the destabilized medial meniscus (DMM) operation was performed to establish the OA mice model. After knee intra-articular injection with OB for 8 weeks, the mice's knee joints were obtained for subsequent histological staining and analysis.
Results: OB reversed the expression level of anabolic-related proteins (Aggrecan and Collagen II) and catabolic-related (MMP3, MMP13, and ADAMTS5) in IL-1β-induced chondrocytes. Mechanistically, OB suppressed the inflammatory response stimulated by IL-1β, as the inflammation-related (iNOS, COX-2, TNF-α, IL-6, and IL-1β) markers were downregulated after the administration of OB in IL-1β-induced chondrocytes. Besides, the activation of PI3K/AKT/mTOR signaling pathway induced by IL-1β could be inhibited by OB. Additionally, the autophagy process impaired by IL-1β could be rescued by OB. What's more, the introduction of 3-MA to specifically inhibit the autophagic process impairs the protective effect of OB on cartilage. In vivo, histological staining revealed that intra-articular injection of OB attenuated the cartilage degradation, as well as reversed the expression level of anabolic and catabolic-related proteins such as Aggrecan, Collagen II, and MMP13 induced in DMM-induced OA models.
Conclusions: The study verified that OB exhibited the chondroprotective effect by anti-inflammatory, inhibiting the PI3K/AKT/mTOR signaling pathway, and enhancing the autophagy process, indicating that OB might be a promising agent for the treatment of OA.
Keywords: PI3K/AKT/mTOR; autophagy; chondrocytes; inflammation; oroxin B; osteoarthritis.
Copyright © 2022 Lu, He, Zhang, Wang, Cheng, Lv, Yuan, Guo, You, Chen and Hu.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures







References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

