Copper-promoted direct sulfenylation of C1-H bonds in 4-aryl pyrrolo[1,2- a]quinoxalines
- PMID: 36540248
- PMCID: PMC9727684
- DOI: 10.1039/d2ra05078a
Copper-promoted direct sulfenylation of C1-H bonds in 4-aryl pyrrolo[1,2- a]quinoxalines
Abstract
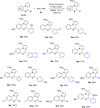
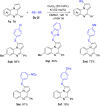
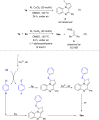
Methods for direct functionalization of C(sp2)-H bonds in pyrrolo[1,2-a]quinoxalines have witnessed emerging development over the last decade. Herein we report a new tactic to afford a selective sulfenylation of 4-aryl pyrrolo[1,2-a]quinoxalines with diaryl disulfides. The reactions proceeded in the presence of a copper catalyst and potassium iodide promoter. Functionalities including nitro, ester, amide, methylthio, and halogen groups were all tolerated. Our method offers a convenient route to obtain highly substituted pyrrolo[1,2-a]quinoxalines-based thioethers in moderate to good yields.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures

References
-
-
Selected examples:
- Guillon J. Forfar I. Mamani-Matsuda M. Desplat V. Saliège M. Thiolat D. Massip S. Tabourier A. Léger J.-M. Dufaure B. Haumont G. Jarry C. Mossalayi D. Bio. Med. Chem. 2007;15:194. doi: 10.1016/j.bmc.2006.09.068. - DOI - PubMed
- de Lucio H. García-Marín J. Sánchez-Alonso P. García-Soriano J. C. Toro M. Á. Vaquero J. J. Gago F. Alajarín R. Jiménez-Ruiz A. Eur. J. Med. Chem. 2022;227:113915. doi: 10.1016/j.ejmech.2021.113915. - DOI - PubMed
- Biswas C. Krishnakanth K. N. Lade J. J. Chaskar A. C. Tripathi A. Chetti P. Soma V. R. Raavi S. S. K. Chem. Phys. Lett. 2019;730:638. doi: 10.1016/j.cplett.2019.06.062. - DOI
-
-
-
Selected reviews:
- Kalinin A. A. Islamova L. N. Fazleeva G. M. Chem. Heterocycl. Compd. 2019;55:584. doi: 10.1007/s10593-019-02501-w. - DOI
- Le H. X. Nguyen T. T. ChemistrySelect. 2022;7:e202200166.
-
-
- Yang Z. He J. Wei Y. Li W. Liu P. Zhao J. Wei Y. Org. Biomol. Chem. 2020;18:9088. doi: 10.1039/D0OB01818J. - DOI - PubMed
- Li Y. Yang Z. Liu Y. Liu Y. Gu Y. Liu P. Mol. Catal. 2021;511:111747. doi: 10.1016/j.mcat.2021.111747. - DOI
- Liu Y. Wei Y. Yang Z. Li Y. Liu Y. Liu P. Org. Biomol. Chem. 2021;19:5191. doi: 10.1039/D1OB00759A. - DOI - PubMed
- Le H. X. Hoang T. N. B. Tran T. H. Nguyen C. T. D. Chiem L. N. T. Phan N. T. S. Nguyen T. T. Tetrahedron Lett. 2021;67:152879. doi: 10.1016/j.tetlet.2021.152879. - DOI
- Yang Z. He J. Wei Y. Li W. Liu P. Org. Biomol. Chem. 2020;18:3360. doi: 10.1039/D0OB00494D. - DOI - PubMed
- Li Y. Liu Y. Hao D. Li C. Liu Y. Gu Y. Vaccaro L. Liu P. Tetrahedron. 2022;105:132610. doi: 10.1016/j.tet.2021.132610. - DOI
- Hao D. Yang Z. Liu Y. Li Y. Liu Y. Liu P. J. Mol. Struct. 2022;1267:133636. doi: 10.1016/j.molstruc.2022.133636. - DOI - PubMed
- Hao D. Yang Z. Liu Y. Li Y. Li C. Gu Y. Vaccaro L. Liu J. Liu P. Org. Biomol. Chem. 2022;20:847. doi: 10.1039/D1OB02248B. - DOI - PubMed
-
-
Selected recent reviews:
- Kaiser D. Klose I. Oost R. Neuhaus J. Maulide N. Chem. Rev. 2020;119:8701. doi: 10.1021/acs.chemrev.9b00111. - DOI - PMC - PubMed
- Annamalai P. Liu K.-C. Badsara S. S. Lee C.-F. Chem. Rec. 2021;21:3674. doi: 10.1002/tcr.202100133. - DOI - PubMed
- Azzi E. Lanfranco A. Moro R. Deagostino A. Renzi P. Synthesis. 2021;53:3440. doi: 10.1055/a-1509-5541. - DOI - PubMed
-
-
-
Selected recent examples:
- Gandeepan P. Koeller J. Ackermann L. ACS Catal. 2017;7:1030. doi: 10.1021/acscatal.6b03236. - DOI
- Kajiwara R. Takamatsu K. Hirano K. Miura M. Org. Lett. 2020;22:5915. doi: 10.1021/acs.orglett.0c02012. - DOI - PubMed
- Kathiravan S. Anaspure P. Zhang T. Nicholls I. A. Org. Lett. 2021;23:3331. doi: 10.1021/acs.orglett.1c00829. - DOI - PMC - PubMed
- Vásquez-Céspedes S. Ferry A. Candish L. Glorius F. Angew. Chem., Int. Ed. 2015;54:5772. doi: 10.1002/anie.201411997. - DOI - PubMed
- Song Z. Ding C. Wang S. Dai Q. Sheng Y. Zheng Z. Liang G. Chem. Commun. 2020;56:1847. doi: 10.1039/C9CC09001K. - DOI - PubMed
-
LinkOut - more resources
Full Text Sources

