Clinicopathological findings, prognosis, and Epstein-Barr virus infection in rheumatoid arthritis patients with other iatrogenic immunodeficiency-associated T- and NK-cell lymphoproliferative disorders
- PMID: 36544095
- PMCID: PMC9773610
- DOI: 10.1186/s12885-022-10358-0
Clinicopathological findings, prognosis, and Epstein-Barr virus infection in rheumatoid arthritis patients with other iatrogenic immunodeficiency-associated T- and NK-cell lymphoproliferative disorders
Abstract
Background: Other iatrogenic immunodeficiency-associated (OIIA) T- and natural killer (NK)-cell lymphoproliferative disorders (TNK-LPDs) are rare in patients with rheumatoid arthritis (RA).
Methods: We investigated the clinicopathological characteristics, Epstein-Barr virus (EBV) infection, genetic findings, therapeutic response, and prognostic factors in 21 RA patients with OIIA TNK-LPDs and compared these with those of 39 with OIIA B-cell LPDs (B-LPDs) and 22 with non-OIIA B-LPDs.
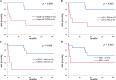
Results: Immunohistologically, 11 patients (52%) showed CD4+ T-LPDs, and 7 had a T follicular helper (TFH) phenotype. The other nine patients (43%) showed CD8+ T-LPDs, and the remaining one (5%) had features of CD3+ CD4- CD8- nasal type TNK-cell lymphoma. CD30+, p53+, and CMYC+ atypical lymphocytes were identified in seven (33%), eight (38%), and five (24%) patients, respectively. In situ hybridisation detected EBV-encoded RNA (EBER) + large atypical lymphocytes in five patients (24%). Nine of 17 patients (53%) showed clonal peaks of TCRγ by polymerase chain reaction. Withdrawal of MTX and biologic drugs was effective in 12 patients (57%), and 8 (38%) received chemotherapies. Two patients with TFH+ or EBV+ CD4+ CD30+ large cell peripheral T-cell lymphoma, one with CD8+ systemic anaplastic large cell lymphoma, and two with systemic EBV+ CD8+ T-cell lymphoma of childhood showed a lethal progressive clinical course within 13 months. Moreover, > 500 U/L LDH, large atypical lymphocytes, expression of CD30, p53, and CMYC, and EBER+ atypical lymphocytes were significantly poor prognostic factors for overall survival (p < 0.05). Median interval from RA onset to OIIA TNK-LPDs was 72 months, which was shorter than 166 months in OIIA B-LPDs (p = 0.003). EBV+ atypical and reactive lymphocytes were frequently found in 15 patients with OIIA TNK-LPDs (71%), in 27 with OIIA B-LPDs (69%), and only in 3 with non-OIIA B-LPDs (14%).
Conclusions: OIIA TNK-LPDs occurred in early phase of RA, compared with OIIA B-LPDs, and occasionally showed a lethal progressive clinical course. Detection of OIIA TNK-LPD patients with poor prognostic factors is necessary. EBV infection in immunosuppressed patients due to persistent RA, MTX, and biologic drugs may play a role in forming the tumour microenvironment and lymphomagenesis of TNK-LPDs.
Keywords: Epstein–Barr virus; Iatrogenic immunodeficiency; Methotrexate; Rheumatoid arthritis; T cells, NK cells, lymphoproliferative disorders.
© 2022. The Author(s).
Conflict of interest statement
The authors declare they have no significant relationships with, or financial interests in, any commercial activities pertaining to this article.
Figures


Similar articles
-
Primary central nervous system other iatrogenic immunodeficiency-associated lymphoproliferative disorders presenting as extraosseous plasmacytoma with a progressive clinical course: A case report and literature review.Neuropathology. 2023 Apr;43(2):151-157. doi: 10.1111/neup.12863. Epub 2022 Sep 5. Neuropathology. 2023. PMID: 36065518 Review.
-
Clinicopathological analyses in patients with other iatrogenic immunodeficiency-associated lymphoproliferative diseases and rheumatoid arthritis.Leuk Lymphoma. 2012 Apr;53(4):616-23. doi: 10.3109/10428194.2011.625101. Epub 2011 Nov 25. Leuk Lymphoma. 2012. PMID: 21933041
-
Diffuse Large B-cell Lymphoma with Adrenal Involvement Presenting as Other Iatrogenic Immunodeficiency-associated Lymphoproliferative Disorders: Sustained Remission with Methotrexate Termination Alone in Two Cases.Intern Med. 2023 Feb 15;62(4):601-604. doi: 10.2169/internalmedicine.0130-22. Epub 2022 Jul 5. Intern Med. 2023. PMID: 35793959 Free PMC article.
-
Case Report: Composite Angioimmunoblastic T-Cell Lymphoma and Epstein-Barr Virus-Positive B-Cell Lymphoproliferative Disorder as Other Iatrogenic Immunodeficiency-Associated Lymphoproliferative Disorders.Front Med (Lausanne). 2020 Dec 23;7:625442. doi: 10.3389/fmed.2020.625442. eCollection 2020. Front Med (Lausanne). 2020. PMID: 33425968 Free PMC article.
-
Other Iatrogenic Immunodeficiency-Associated Lymphoproliferative Disorders with a T- or NK-cell phenotype.J Clin Exp Hematop. 2019;59(2):56-63. doi: 10.3960/jslrt.19013. J Clin Exp Hematop. 2019. PMID: 31257346 Free PMC article. Review.
Cited by
-
Shared Pathogenicity Features and Sequences between EBV, SARS-CoV-2, and HLA Class I Molecule-binding Motifs with a Potential Role in Autoimmunity.Clin Rev Allergy Immunol. 2023 Aug;65(2):206-230. doi: 10.1007/s12016-023-08962-4. Epub 2023 Jul 28. Clin Rev Allergy Immunol. 2023. PMID: 37505416 Review.
References
-
- Swerdlow S, Campo E, Harris NL, et al., editors. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues, revised. 4. Lyon: IARC Press; 2017.
-
- Mariette X, Cazals-Hatem D, Warszawki J, Liote F, Balandraud N, Sibilia J. and the investigators of the Club Rhumatismes et Inflammation. Lymphomas in rheumatoid arthritis patients treated with methotrexate: A 3-year prospective study in France. Blood. 2002;99(11):3909–3915. doi: 10.1182/blood.v99.113909. - DOI - PubMed
-
- Yamada K, Oshiro Y, Okamura S, Fujisaki T, Kondo S, Nakayama Y, Suematsu E, Tamura K, Takeshita M. Clinicopathological characteristics and rituximab addition to cytotoxic therapies in patients with rheumatoid arthritis and methotrexate-associated large B lymphoproliferative disorders. Histopathol. 2015;67(1):70–80. doi: 10.1111/his.12627. - DOI - PubMed
-
- Kurita D, Miyoshi H, Ichikawa A, Kato K, Imaizumi Y, Seki R, Sato K, Sasaki Y, Kawamoto K, Shimono J, et al. Methotrexate-associated lymphoproliferative disorders in patients with rheumatoid arthritis clinicopathologic features and prognostic factors. Am J Surg Pathol. 2019;43(7):869–884. doi: 10.1097/PAS.0000000000001271. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

